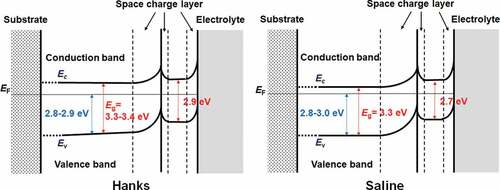
Figures & data
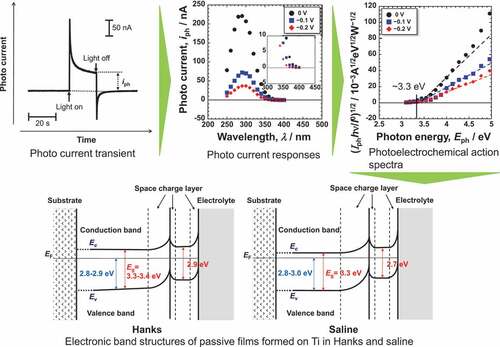
Figure 2. Example of photocurrent transient generated with the light on and off for passive films on Ti.
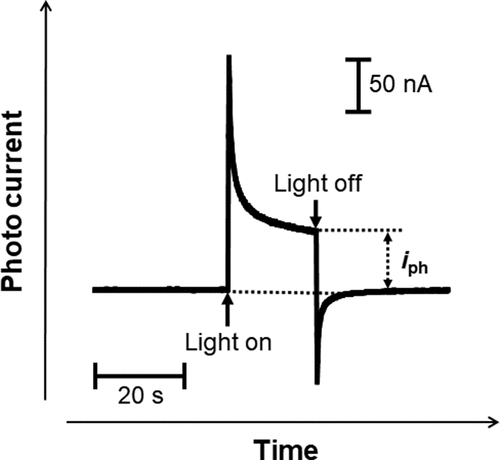
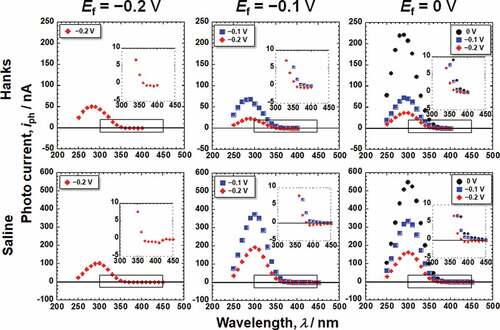
Figure 4. Photoelectrochemical action spectra calculated from the steady photocurrent in Hanks shown in .
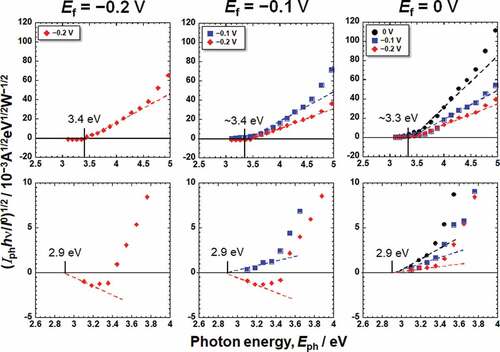
Figure 5. Photoelectrochemical action spectra calculated from the steady photocurrent in saline shown in .
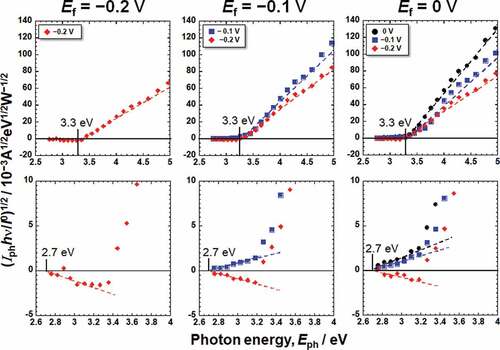
Figure 6. (a) Ti 2p, (b) O 1s, (c) Ca 2p, and (d) P 2p electron energy region spectra obtained from Ti after polarization at 0 V in Hanks for 1 h.
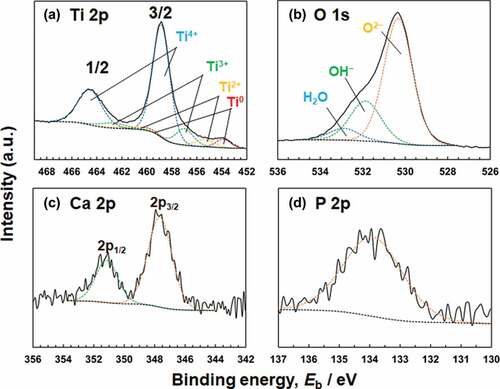
Figure 7. [OH−]/[O2−] ratios calculated form O 1s electron energy region spectra of Ti before and after polarization at each film formation potential, Ef (n = 3).
![Figure 7. [OH−]/[O2−] ratios calculated form O 1s electron energy region spectra of Ti before and after polarization at each film formation potential, Ef (n = 3).](/cms/asset/80622094-3f44-4f31-a02f-21f279e229d3/tsta_a_2066960_f0007_oc.jpg)
Table 1. Relative concentrations of elements, [Ca]/[p] ratios, and thickness of the passive film formed on Ti (n = 3)