Figures & data
Table 1. Main characteristics of the currently available α-particle-emitting radionuclides.
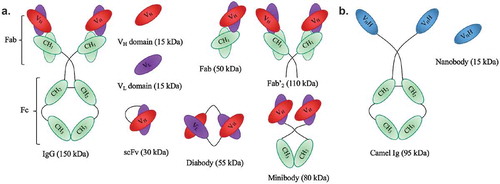
Table 2. Vehicles used in targeted α-particle therapy in preclinical and clinical settings.
![Figure 2. Nanobodies possess numerous advantageous characteristics, including their high antigen specificity (a) and high tumor targeting potential (b). a. 99mTc-labeled-nanobody targeting the complement receptor of the Ig superfamily, CRIg, expressed on Kupffer cells in the liver. 3D-rendered SPECT/micro-CT images of naive wild-type (A.1.) and CRIg−/− mice (A.2.) 1 h after intravenous injection of 99mTc-labeled-nanobody. Representative images for 3 mice per group are shown. Figures adapted with permission from.[Citation115] b. Dosimetry calculation of untagged 177Lu-DTPA-anti-HER2 nanobody coinfused with 150 mg/kg Gelofusin, in HER2pos tumor xenografted mice. Radiolabeling of nanobodies is characterized by significant retention of radioactivity at the kidneys, due to the charged-based aspecific tubular reuptake after glomerular filtration. Figure adapted with permission from.[Citation109].](/cms/asset/3d9ef9ac-46d8-4405-bd54-ccf7599fa7d7/iebt_a_1185412_f0002_c.jpg)
![Figure 3. Diagnostic tumor imaging using 68Ga-HER2-nanobody in patients with HER2pos-breast cancer. a. Time-activity curve of total blood activity, expressed in % of injected activity (%IA) (n=20). b. Fusion PET/CT images of the uptake of 68Ga-HER2-nanobody in breast carcinoma lesions. (B.1.) Patient with the highest tracer uptake (SUVmean 11.8) in a primary breast carcinoma. (B.2.) Patient with moderate tracer uptake in the left breast, which is easily discernable from background (SUVmean 4.9). (B.3.) Patient with invaded lymph nodes in the mediastinum and left hilar region. Lesions are indicated by red arrows. Figures are adapted with permission from.[Citation110].](/cms/asset/21ddf3b1-eb42-4799-9ce3-0564a98dd92f/iebt_a_1185412_f0003_c.jpg)
