Figures & data
Figure 1. T-cell co-expressing 4ab and T1E28z interacting with ErbB dimers expressed on a tumor cell.
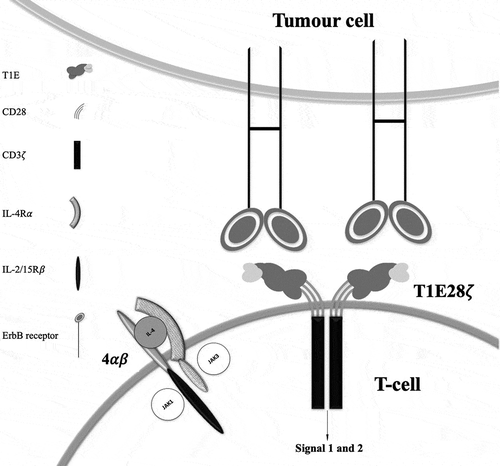
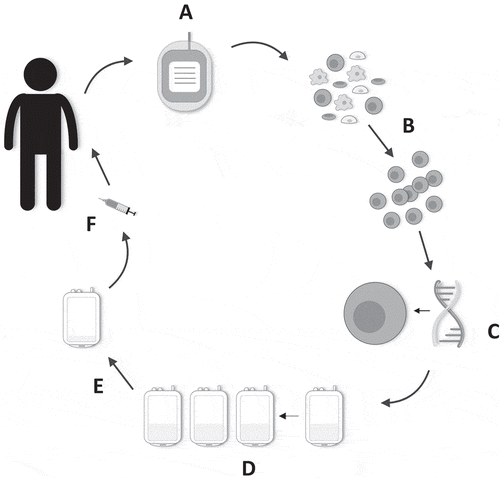
Figure 2. Manufacture of T4 immunotherapy. (a) 40–120 mL of whole blood is harvested from the patient. (b) Patient-derived peripheral blood mononuclear cells are isolated and activated. (c) Gene transfer is performed after 48 h. (d) Preferential expansion and enrichment of T4+ T-cells ex vivo is achieved with the addition of IL-4 to the cell culture bags. (e) Directly prior to administration, the cell culture is volume-reduced. (f) Cells are contained in a final 1–4 mL dosing volume and drawn up into a syringe. The dose is administered intratumorally using ultrasound guidance where required.