Figures & data
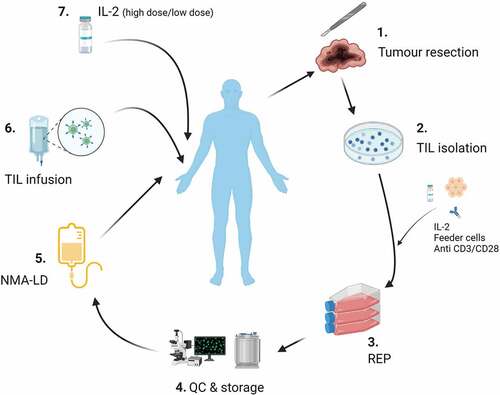
Figure 1. Tumor infiltrating lymphocyte (TIL) manufacture/administration. 1) Surgical resection of tumor tissue. 2) TIL isolation. 3) Rapid expansion protocol (REP): This process, originally developed by the Rosenberg team at the NCI, involves the use of IL-2, feeder cells and anti CD3/CD28 monoclonal antibodies to produce a therapeutic TIL product. 4) Quality control (QC) and storage. 5) Non myeloablative- lymphodepletion (NMA-LD). 6) TIL infusion. 7) Adjuvant IL-2: Dose and route of administration vary between clinical trials. Image created with Biorender.Com.