Figures & data
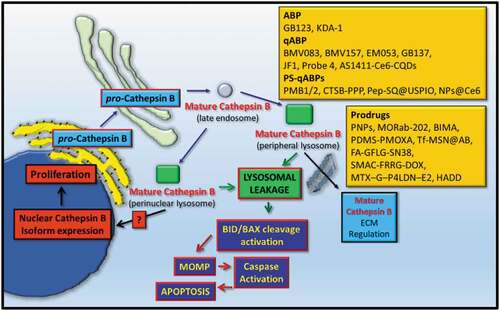
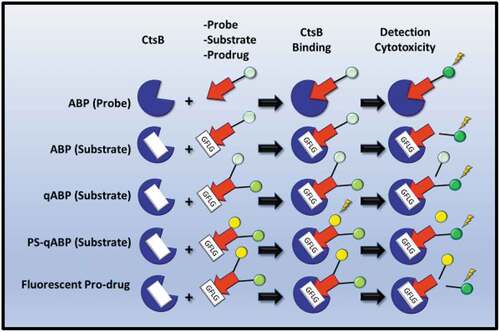
Table 1. Activity-Based Probes (ABPs) specific for detecting cathepsin proteases are highlighted. ABPs were tested for specific cathepsin protease detection using Gel-Labeling assays, cellular imaging approaches or in whole animal systems. The diseases that the ABPs have been tested for are also highlighted. NA-Not Applicable; Y-Yes; Mac-Macrophages; OA-Osteoarthritis; BC-Breast Cancer; K-Kidney; M-Mouse; AS-Atherosclerosis; KidC-Kidney Cancer.
Table 2. Quenched Activity-Based Probes (qABPs) specific for detecting cathepsin proteases are highlighted. qABPs were tested for specific cathepsin protease detection using Gel-Labeling assays, cellular imaging approaches or in whole animal systems. The diseases that the qABPs have been tested for are also highlighted. NA-Not Applicable; Y-Yes; BMM-Bone Marrow Macrophages; Mac-Macrophages; M-Mouse; BC-Breast Cancer; AS-Atherosclerosis; PrC-Prostate Cancer; CerC-Cervical Cancer; KidC-Kidney Cancer.
Table 3. Photosensitization quenched Activity-Based Probes (PS-qABPs) carrying specific cleavage sequences for Cathepsin B (CtsB) are highlighted. PS-qABPs were tested for specific CtsB detection in cells in vitro, and in vivo using whole animal systems. IC50 concentrations are highlighted as are their abilities to inhibit tumor proliferation. The diseases that the qABPs have been tested for are also highlighted. NA-Not-Applicable; M-Mouse; H-Human; Lun-Lung; Liv-Liver; PTX-Paclitaxel; TNBC-Triple-Negative Breast Cancer; BladC-Bladder Cancer; Xeno-Xenografts.
Table 4. Selected Prodrugs are highlighted along with specific cleavage sequences for Cathepsin B (CtsB). PS-qABPs drugs were tested for their inhibitory effects in vitro cellular systems and in whole-animal systems. DOX-Doxorubicin; MIS-Minimal Inhibitory Sequence; M-Mouse; NA-Not-Applicable; BC-Breast Cancer; FibS-Fibrosarcoma; TNBC-Triple-Negative Breast Cancer; ColC-Colon cancer; Card-Cardiomyocytes; PanC-Pancreatic Cancer; PrC-Prostate Cancer; LuC-Lung Cancer; Pan-Pancreatic Epithelial; Ova-Ovarian Cancer; CerC-Cervical Cancer; GasC-Gastrointestinal Cancer; Endo-Endothelial; LivC-Liver Cancer; NSCLC-Non-Small Cell Lung Cancer.