Figures & data
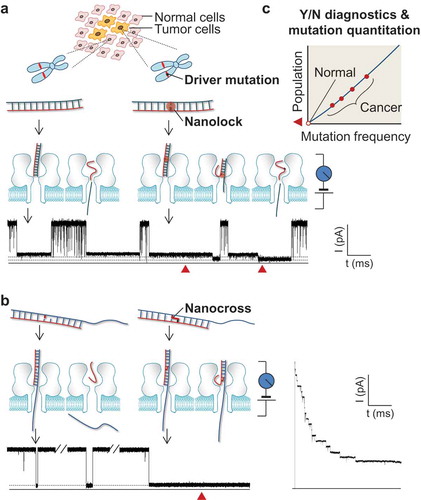
Figure 1. Single-molecule detection of cancer driver mutation by using nanolock-nanapore and nanocross-nanopore sensors. (a) Nanolock-nanopore detection of the BRAF V600E mutant allele in tumor tissue DNA sample; (b) Nanocross-nanopore detection of the BRAF V600E mutant DNA sequence. In both methods, the non-covalent nanolock (a) or covalent nanocross (b) is constructed only on the mutant•probe DNA complex. Their special molecular configuration changes in the nanopore produce a unique ion current signature (current blockade marked by red solid triangle), which serves as a fingerprint of a single mutant DNA molecule. For the nanocross method (b), the unique long-duration blockade by the nanocross enables multi-nanopore simultaneous detection of mutant DNA; (c) Identification of mutant fingerprints enables Yes/No early cancer diagnostics, and counting of mutant fingerprints allows quantitation of mutant frequency for personalized treatment. This figure was adapted with permission from (Wang Y, Tian K, Shi R et al. Nanolock-Nanopore Facilitated Digital Diagnostics of Cancer Driver Mutation in Tumor Tissue. ACS sensors, 2(7), 975–981 (2017)). Copyright (2017) American Chemical Society; adapted with permission from (Zhang X, Price NE, Fang X, Yang Z, Gu LQ, Gates KS. Characterization of Interstrand DNA-DNA Cross-Links Using the alpha-Hemolysin Protein Nanopore. ACS nano, 9(12), 11812–11819 (2015)). Copyright (2015) American Chemical Society; adapted from (Nejad MI, Shi R, Zhang X, Gu LQ, Gates KS. Sequence-Specific Covalent Capture Coupled with High-Contrast Nanopore Detection of a Disease-Derived Nucleic Acid Sequence. ChemBioChem, 18(14), 1383–1386 (2017)) with permission of John Wiley and Sons, Copyright (2017).