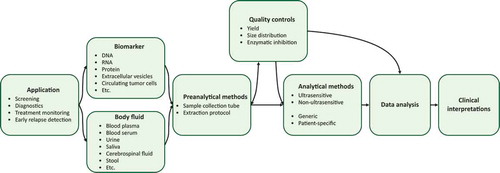
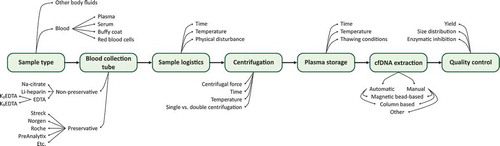
Figures & data
Table 1. Overview of comparative preanalytical studies