Open access
691
Views
0
CrossRef citations to date
0
Altmetric
Original Research
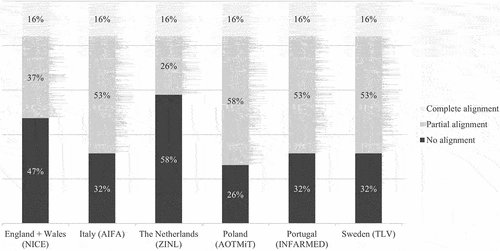
Differences in evidentiary requirements for oncology drug effectiveness assessments among six European health technology assessment bodies — can alignment be improved?
Sharon Woltersa Asc Academics B.V, Groningen, The Netherlands;b Department of Health Sciences, University of Groningen, University Medical Center, Groningen, Groningen, The NetherlandsCorrespondence[email protected]
View further author information
, View further author information
Lisa A de Jongb Department of Health Sciences, University of Groningen, University Medical Center, Groningen, Groningen, The Netherlands https://orcid.org/0000-0001-8814-0670View further author information
https://orcid.org/0000-0001-8814-0670View further author information
Christel Jansenc Vintura Consultancy, Baarn, the Netherlands;d Inbeoo Ltd (current), London, The UKView further author information
, Frank GA Jansmane Department of Clinical Pharmacy, Deventer Teaching Hospital, Deventer, the Netherlands;f Department of Pharmacotherapy, Pharmacoepidemiology and Pharmacoeconomics, Groningen, Research Institute of Pharmacy (GRIP), University of Groningen, Groningen, the NetherlandsView further author information
& Maarten J Postmab Department of Health Sciences, University of Groningen, University Medical Center, Groningen, Groningen, The Netherlands;g Department of Economics, Econometrics & Finance, University of Groningen, Groningen, the Netherlands;h Center of Excellence in Higher Education for Pharmaceutical Care Innovation, Padjadjaran University, Bandung, IndonesiaView further author information
Pages 251-265
|
Received 24 Jun 2023, Accepted 19 Sep 2023, Published online: 28 Sep 2023
Related research
People also read lists articles that other readers of this article have read.
Recommended articles lists articles that we recommend and is powered by our AI driven recommendation engine.
Cited by lists all citing articles based on Crossref citations.
Articles with the Crossref icon will open in a new tab.