Figures & data
Table 1. Base case analysis – Methods – BIM main parameters – I.
Table 2. Base case analysis – Methods – BIM main parameters – II.
Table 3. Base case analysis – Results – Cost and saving for the INHS – I – Scenario without inebilizumab (€2023)a.
Table 4. Base case analysis – Results – Cost and saving for the INHS – II – Scenario with inebilizumab (€2023)a.
Figure 1. Base case analysis - Number of NMOSD attacks - Scenario without inebilizumab (Panel A), scenario with inebilizumab (Panel B) and difference between the two scenarios (Panel C)
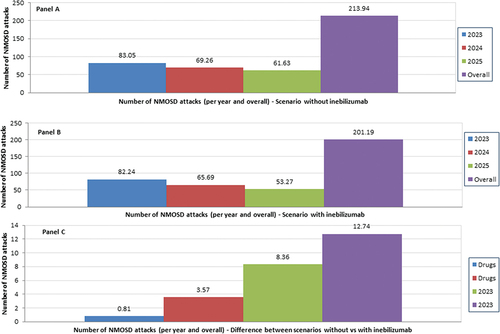
Figure 2. One-way sensitivity analysis - Results concerning the first 10 parameters of the BIM that causes the largest variations in the base case INHS saving due to inebilizumab availability (€8,373,125.13) (€2023). a
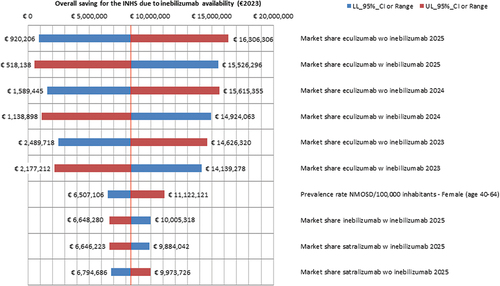
Table 5. Scenario sensitivity analysis – Results (€2023).
Figure 3. Threshold value analysis – I - Effect of inebilizumab market share variation on the base case INHS saving due to inebilizumab availability (€8,373,125.13) (€2023).a
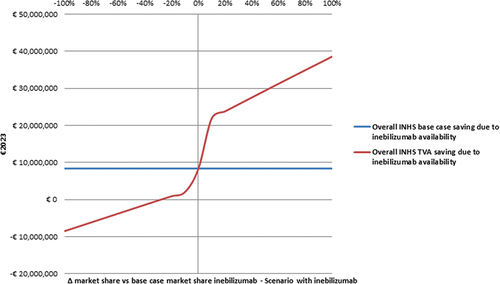
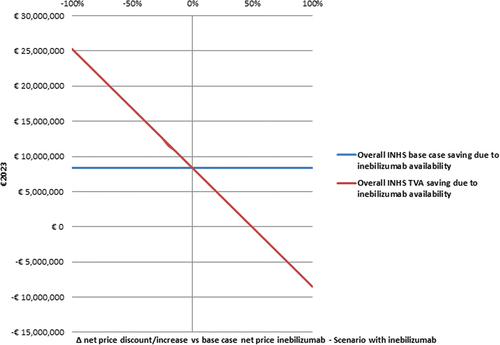
Figure 4. Threshold value analysis – II - Effect of inebilizumab net price variation on the base case INHS saving due to inebilizum abavailability (€8,373,125.13) (€2023).