Figures & data
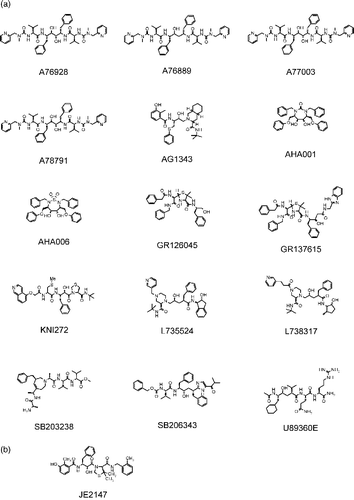
Table I. HIV-1 protease inhibitors used in this study.
Table II. Energy (kcal/mol) of the complexes with protonation at Asp25 or Asp25′a.
Table III. Calculated parameters for linear regression modeling.
Table IV. Statistical results of linear regression models.
Table V. Experimental, calculated, and predicted free-energy differences.