Figures & data
Scheme 1 Synthesis of glutathione analogs. Synthesis of tripeptides ASC-I-74A, ASC-I-74B, ASC-I-75A, ASC-I-74B: (i) N-α-benzyloxycarbonyl-L-glutamic acid α-benzyl 1, N-hidroxysuccinimide, dry dioxane, DCC, 0°C, 12 h, 98%; (ii) R = L-Leu or L-Val, H2O, Et3N, ester 2, THF, rt, 12 h, 76%; (iii) N-α-benzyloxycarbonyl-L-glutamyl(α-benzyl)-L-Leu 3 or L-Val 3′, N-hidroxysuccinimide, dry dioxane, DCC, 0°C, 12 h, 96%; (iv) W = Gly or L-Ala, H2O, Et3N, ester 4 or 4′, THF, rt, 12 h, ASC-I-74A: 85%; ASC-I-74B: 81%; ASC-I-75A: 83%; ASC-I-75B: 76%.
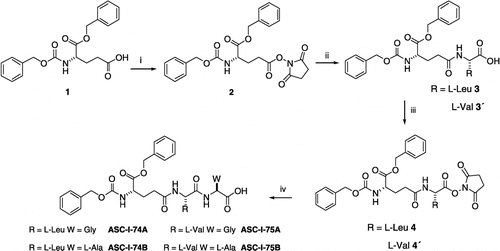
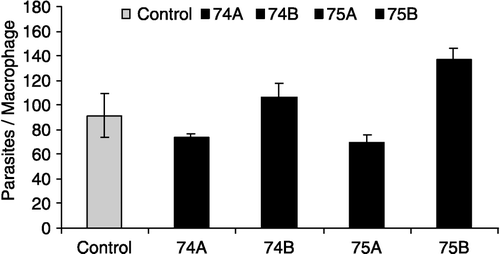
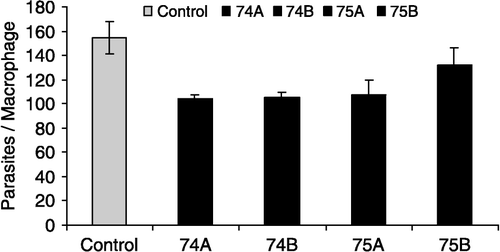
Table I. Effect of glutathione analogs on Leishmania amazonensis infective promastigotes. Parasites were incubated with/without the analogs and after 24 h the LDs50 were calculated.
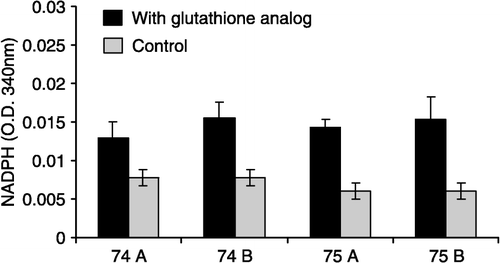
Figure 1 Effect of glutathione analogs on the activity of TR of Leishmania amazonensis infective promastigotes. The enzyme activity was measured by NADPH consumption.