Figures & data
Figure 1 Structures of series 1-3. The aryl substituents are as follows: a: R1 = R2 = H; b: R1 = 4-Cl, R2 = H; c: R1 = 3-Cl, R2 = 4-Cl; d: R1 = 4-CH3, R2 = H; e: R1 = 4-OCH3, R2 = H; f: R1 = 4-F, R2 = H; g: R1 = 4-Br, R2 = H; h: R1 = 2-NO2, R2 = H; i: R1 = 3-NO2, R2 = H; j: R1 = 4-NO2, R2 = H.
Table I. Examination of 2a-j, 3a-j and melphalan against human Molt 4/C8 and CEM T-lymphocytes as well as murine L1210 cells
Table II. A comparison of the relative potency (RP) values of series 1-3a
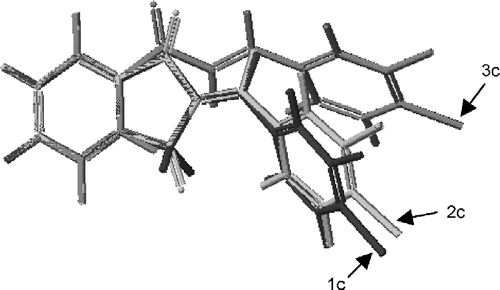
Figure 3 Interatomic distances (d1, d2) and bond angles (Ψ1, Ψ2) measured in order to determine the location of ring B and the C* atom in relation to ring A in 1c, 2c and 3c.
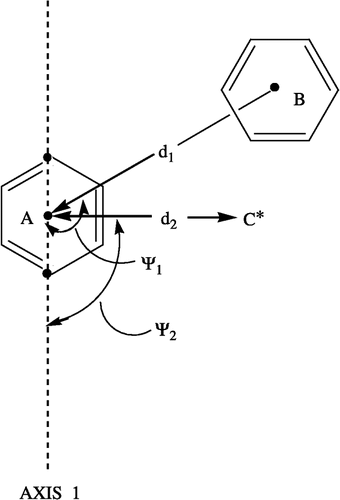
Table III. Various interatomic distances (d1, d2), bond angles (ψ1, ψ2) and a torsion angle (θ1) in 1c, 2c and 3c determined by molecular modeling
Table IV. Charge densities of the olefinic carbon atoms adjacent to aryl ring B