Figures & data
Scheme 1 (i) NaOH (5% aqueous), ether, stirring at RT, 6 h (ii) H2SO4 / ethanol, refluxing, 11 h (iii) NH2-NH2.H2O / ethanol (absolute), refluxing, 9 h (iv) CS2 / KOH, ethanol, refluxing, 16-17 h (v) Et3N, DMAP, CHCl3 (dry), benzyl bromide or p-nitrobenzyl bromide, stirring at 30–70°C, 5–7 h.
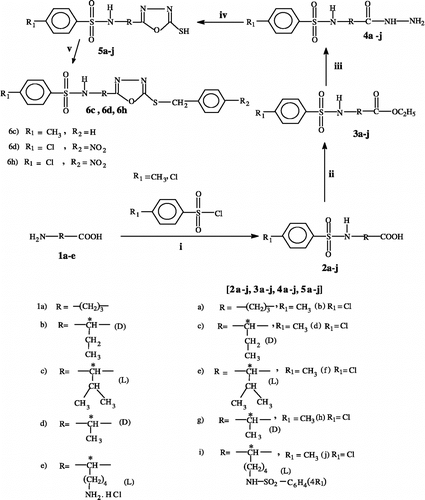
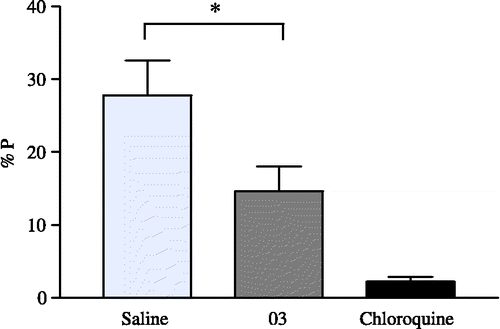
Table I. Effects of compounds on the hem crystallization and hemoglobin proteolysis.