Figures & data
Scheme 1 Synthesis of compounds 1a-l. Reagents: (i) DMTHF, CH3COOH, Δ; (ii) BiCl3 / NaBH4, EtOH; (iii) CO(OCCl3)2, toluene, Δ; (iv) POCl3, Δ; (v) Method A: NaH, HS-CH2-CH2-R3, dioxane; Method B: K2CO3, HS-CH2-CH2-R3, CuI, HOCH2CH2OH, 2-propanol; Method C: t-BuONa, HS-CH2-CH2-R3, CuI, neocuproine, toluene; Method D: t-BuOK, HS-CH2-CH2-R3, Pd(PPh3)4, n-BuOH (vi) CS2, NaOH, EtOH; (vii) NaH, Cl-CH2-CH2-R3, DMF; (viii) HCl, Et2O.
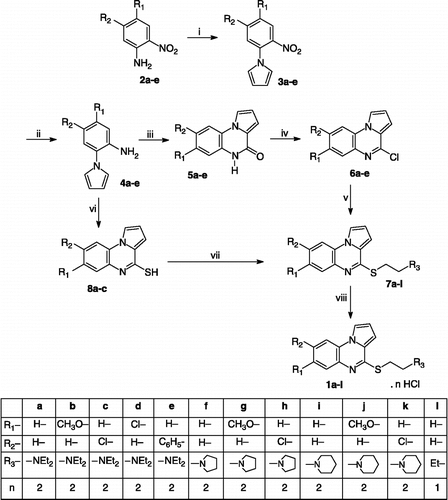
Table I. Optimized reaction conditions for coupling of 6a and N,N-diethylaminoethylthiol.
Scheme 2 Synthesis of compound 1m. Reagents: (i) Method A: NaH, HS-CH2-CH2-N(C2H5)2, dioxane; (ii) HCl, Et2O.
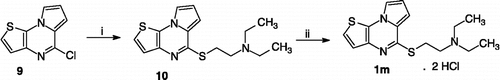
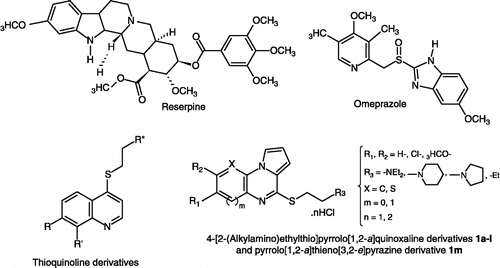
Table II. EPI activities of reserpine, omeprazole, 4-[2-(alkylamino)ethylthio]pyrrolo[1,2-a]quinoxaline derivatives 1a-l and pyrrolo[1,2-a]thieno[3,2-e]pyrazine 1m on the S. aureus strains, SA 1199B and SA-1.