Figures & data
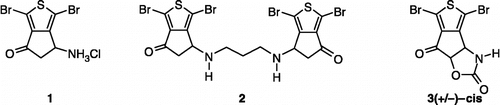
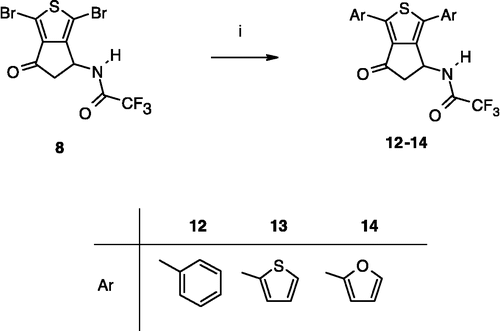
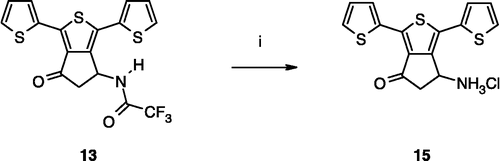
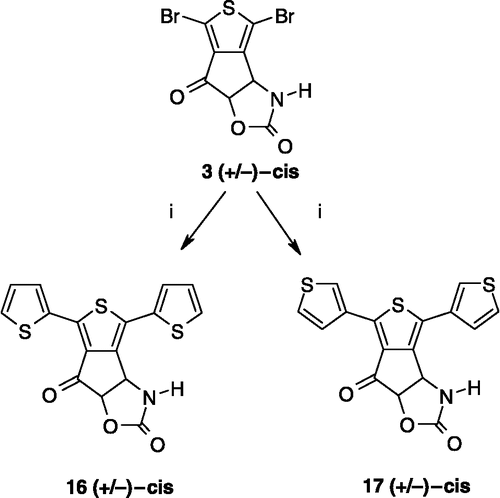
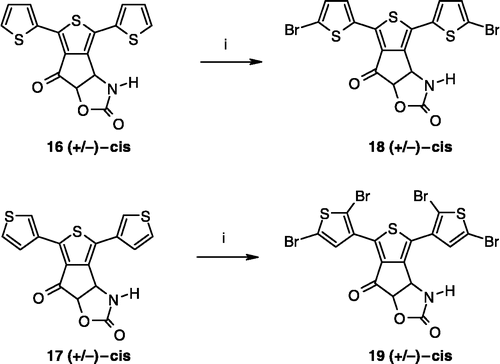
Scheme 1 Synthesis of compounds 3,8. Reagents: (i) AcONH4, (CH2)2CO2H, EtOH; (ii) TFA2O, Et2O; (iii) Br2, CH2Cl2; (iv) SOCl2; (v) AlCl3, CH2Cl2; (vi) Br2, AcOH; (vii) Na2CO3, Me2CO; (viii) HCl gas, Me2CO; (ix) ambient air; (x) (CCl3O)2CO, toluene.
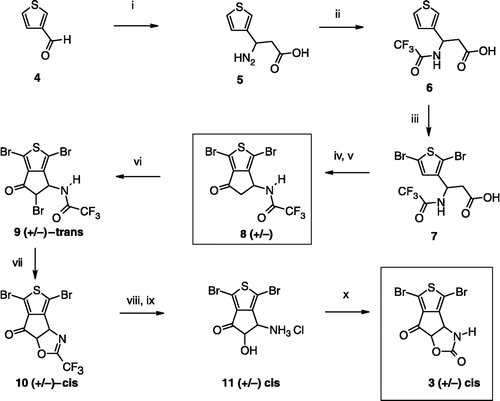
Table I. Cytotoxicity of compounds 15–19 in the NCI's three-cell line one-dose primary anticancer assay, expressed as the percent growth at 10−4 M concentration.
Table II. Cytotoxicity of compounds 1,3,15 in the NCI's in vitro human disease-oriented tumor cell line screening expressed as the log molar drug concentration required for 50% growth inhibition (log GI50)