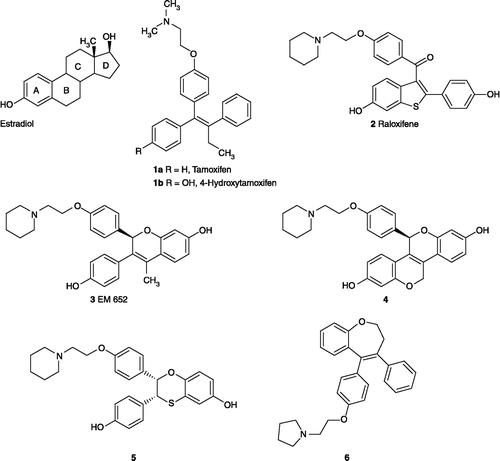
Figures & data
Scheme 1 Synthesis of Benzothiepins 13a–c and 14. Scheme reagents: (a) R = H, F; PPA, 110°C, 4h; R = OMe, Eaton's reagent, 80°C, 2h; (b) i nBuLi, 4-Br-C6H5-OTHP, THF; ii HCI, MeOH.(c) K2CO3, 1-(2-chloroethyl)pyrrolidine hydrochloride, acetone, 24h, 85°C;(d) PyHBr3, CH2 Cl2, 20°, 18th; (e) Pd(PPh3)3, 4-HOC6H5B(OH)2, Na2CO3(aq), THF; (f) BBr3, CH2Cl2.
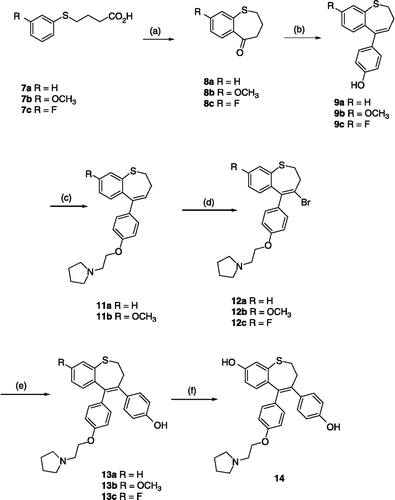
Table I. Antiproliferative effects and estrogen receptor binding affinities for benzothiepin type SERMs.
Scheme 2 Synthesis of Benzothiepin 12c. Scheme reagents: (a) PyHBr3, CH2Cl2, 20°, 18h; (b) K2CO3, 1-(2-chloroethyl)pyrrolidine hydrochloride, acetone, 24h, 85°C.
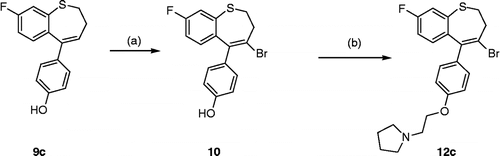
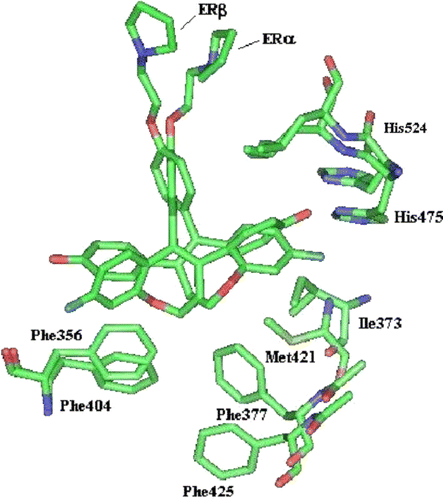
Table II. Summary of key Ligand-Protein contactsa.