Figures & data
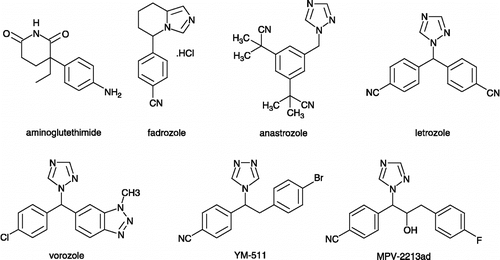
Table I. Inhibition of aromatase by major nonsteroidal inhibitors.
Scheme 1 Synthetic route to 3-azolylmethyl-1H-indoles 11–14. Reagents: (a) see reference [Citation34]; (b) K2CO3, acetone, RCl; (c) NaBH4, methanol; (d) CDI, THF; (e) SDT, CH3CN.
![Scheme 1 Synthetic route to 3-azolylmethyl-1H-indoles 11–14. Reagents: (a) see reference [Citation34]; (b) K2CO3, acetone, RCl; (c) NaBH4, methanol; (d) CDI, THF; (e) SDT, CH3CN.](/cms/asset/5ee80dc2-41e5-4b46-b550-ab7c02ef47ed/ienz_a_265107_f0003_b.gif)
Scheme 2 Synthetic route to 3-(azolylbenzyl)-1H-indoles 30–37. Reagents: (a) see reference [Citation33]; (b) AlCl3, CH2Cl2, ArCOCl; (c) CsCO3, CH3CN, R1Cl; (d) NaBH4, methanol; (e) CDI, THF; (f) SDT, CH3CN.
![Scheme 2 Synthetic route to 3-(azolylbenzyl)-1H-indoles 30–37. Reagents: (a) see reference [Citation33]; (b) AlCl3, CH2Cl2, ArCOCl; (c) CsCO3, CH3CN, R1Cl; (d) NaBH4, methanol; (e) CDI, THF; (f) SDT, CH3CN.](/cms/asset/510b5911-bc41-4fd3-95f4-13c39c3b7ce6/ienz_a_265107_f0004_b.gif)
Table II. Inhibition of P450 arom and P450 17α by 3-(azolylmethyl)-1H-indoles 11–18.
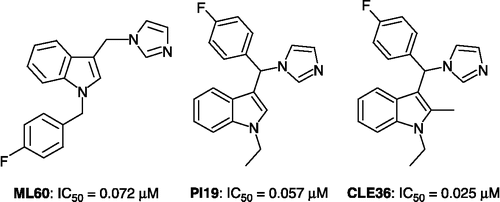
Table III. Inhibition of P450 arom and P450 17α by 3-(α-azolylbenzyl)-1H-indoles 30–37.