Figures & data
Figure 1. The chemical structures of different flavonoid subclasses assayed for their effects on AO activity.
Table I. The effects of some flavonoids on the oxidation of vanillin and phenanthridine catalyzed by guinea pig liver AO and xanthine oxidation catalyzed by guinea pig liver XO.
Table II. IC50 values for the inhibition of guinea pig liver AO and XO by various flavonoids, menadione and allopurinol.
Table III. Kinetic characteristics of the inhibitory activity of flavonoids on the oxidation of vanillin and phenanthridine by guinea pig liver AO.
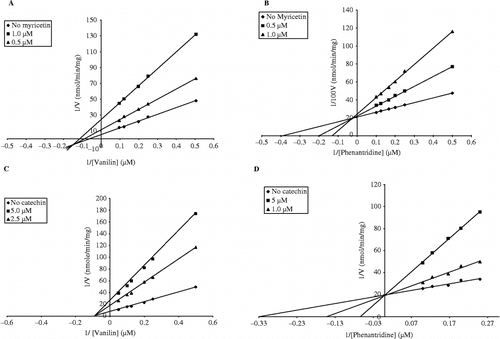
Figure 2. Typical Lineweaver-Burk plots for inhibitory activity of myricetin and catechin on the oxidation of vanillin (A and C, respectively) and phenanthridine (B and D, respectively) catalyzed by guinea pig liver AO. The enzyme assays were performed as described in materials and methods. The data represent the average of 4–8 experiments.