Figures & data
Scheme 1. General scheme for the synthesis of compounds 1–7 and their in vitro Jack bean urease inhibitory activities.
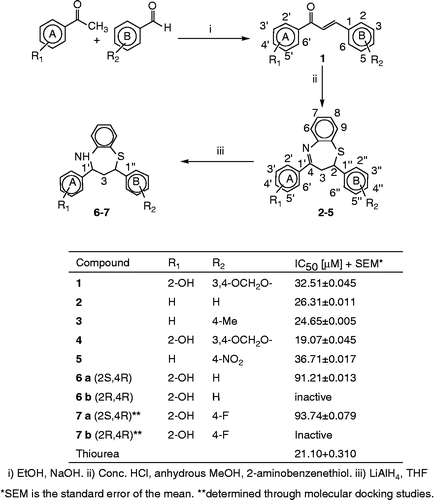
Figure 1. Compound 1 into the active site of urease. The molecule in gray is the ligand with the active site residues. Metals are represented in green.
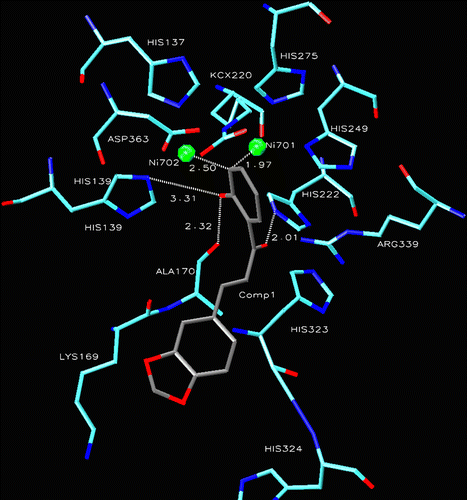
Figure 2. Compound 4 into the active site of urease. The lignad is represented as a ball and stick model.
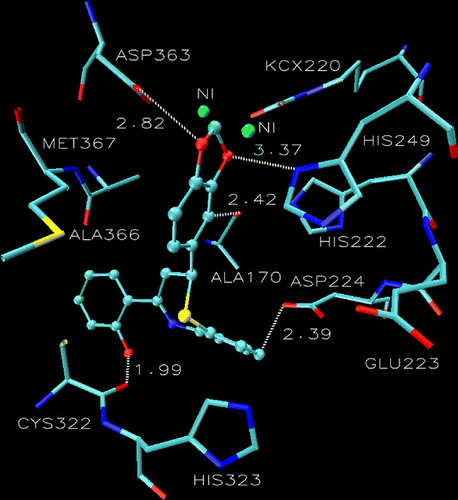
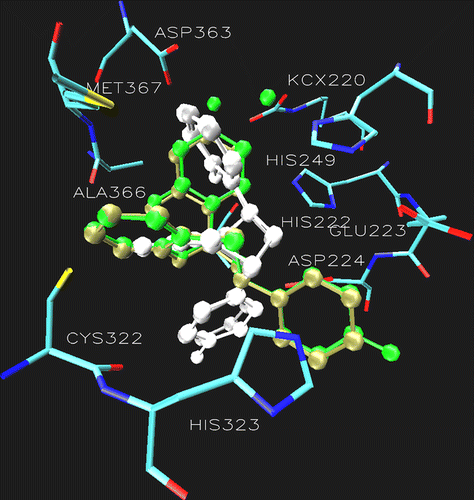
Figure 3. Superimposition of the docking pose of 6a (tan), 6b (gray), 7a (green) 7b (white) showing the difference in the orientation of ring B in the catalytic core of urease. Additionally the active site of urease clearly demonstrating the role of Asp224 and Cys322. The ligands are represented as a ball and stick model.