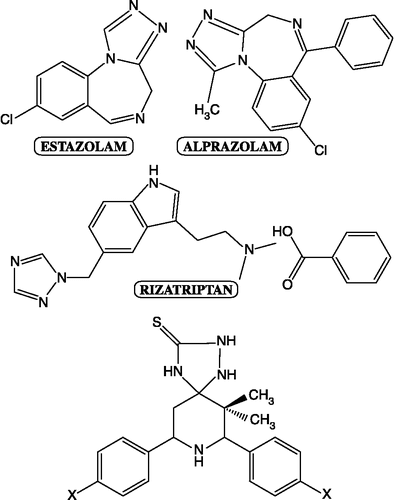
Figures & data
Scheme 2. Synthetic route for the formation of 7,9-diaryl-6,6-dimethyl-1,2,4,8-tetraazaspiro[4,5]-decan-3-thlones.
![Scheme 2. Synthetic route for the formation of 7,9-diaryl-6,6-dimethyl-1,2,4,8-tetraazaspiro[4,5]-decan-3-thlones.](/cms/asset/9361fad8-65ad-401b-b181-a7f48b282dba/ienz_a_318976_f0002_b.gif)
Table I. Physical and analytical data for 7,9-diaryl-6,6-dimethyl-1,2,4,8-tetraazaspiro[4.5]decan-3-thiones (23–27).
Scheme 3. Proposed free radical mechanism for the conversion of thiosemicarbozones to the spiro-1,2,4-triazolidin-3-thiones.
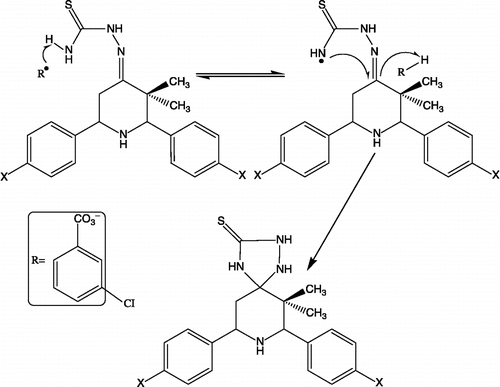
Table II. In vitro antibacterial activity (MIC) values for compounds 23–27.
Table III. In vitro antifungal activity (MIC) values for compounds 23–27.