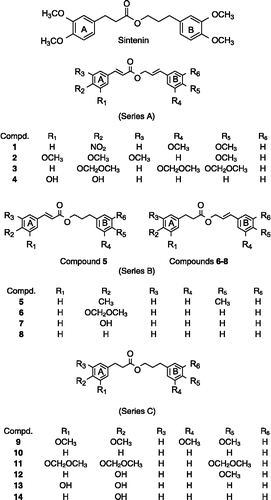
Figures & data
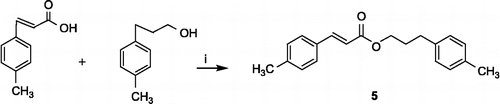
Scheme 1 Synthesis of compound 5. Reagents and conditions: (i) CDI, THF, reflux, 0.5 h; DBU, THF, reflux, 12 h.
Scheme 2 Synthesis of compounds 6–8. Reagents and conditions: (i) DCC, CH2Cl2, rt, 5 min; DMAP, rt, 12 h (ii) 10% HCl, CH3OH, reflux.
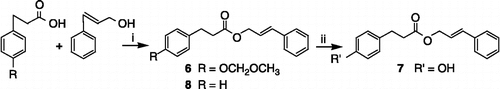
Table I. DPPH radicals scavenging activity by the test compounds.
Figure 2. The inhibitory effects of compounds 4, 13, curcumin, and quercetin on iron-induced lipid peroxidation in rat brain homogenates.
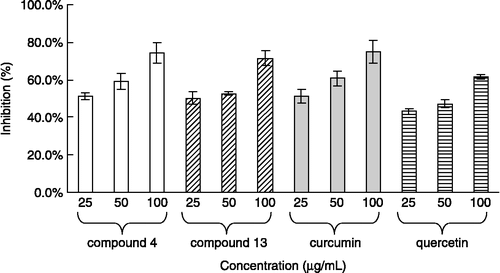
Table II. Ferrous ion chelating ability by the test compound.
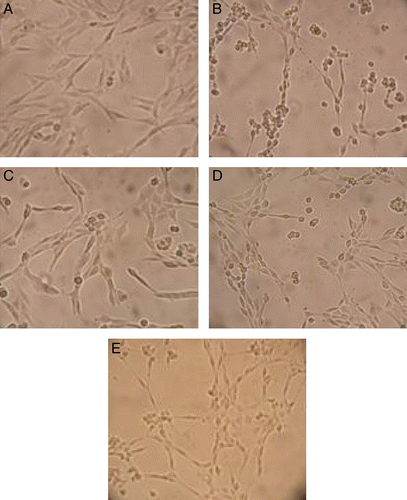
Figure 3. Effects of compounds 4, 13 and positive control on PC 12 cell injury induced by H2O2. (A) PC 12 control cells. (B) PC 12 cells exposed to 600 μM of H2O2 for 3 h. Most of the cells become round shapes. The membrane lesion of cells was also observed. (C, D and E) PC 12 cells were pre-incubated with 20 μg/mL of compounds 4, 13 and quercetin, respectively before exposed to 600 μM H2O2 for 3 h.
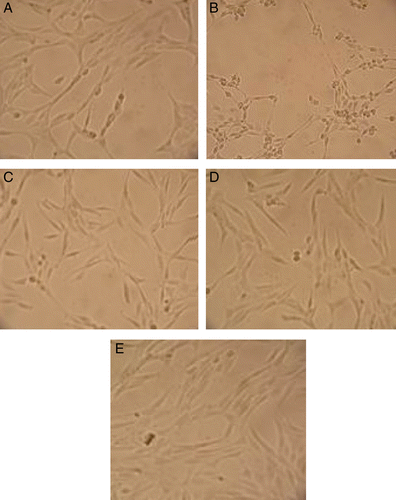
Figure 4. Protective effects of compounds 4, 13, and quercetin against H2![]()
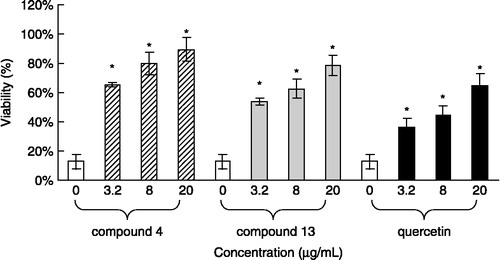
Figure 5. The results of removing test compounds from the pre-incubated medium of PC 12 cells prior to the injury initiated by H2O2. (A) PC 12 control cells. (B) PC 12 cells exposed to 600 μM H2O2 for 3 h. Most of the cells become round shapes. The membrane lesion of cells was also observed. (C, D and E) PC 12 cells were pre-incubated with 20 μg/mL of compounds 4, 13 and quercetin, respectively, and then the compounds were removed from the medium and the cells were exposed to 600 μM H2O2 for 3 h. The cells cultured with compounds 4 and 13 were remarkably improved in cell viabilities and integralities.