Figures & data
Figure 1. Structures of the reference compounds 1 testosterone, 2 dihydrotestosterone, 3 finasteride, 4 dutasteride, 5 4-MA, 6 mibolerone.
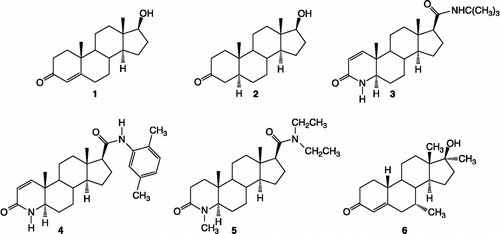
Table I. Weight of prostate glands±standard deviation from animals receiving s.c. treatments for 6 days of differenct compounds.
Figure 2. Synthesis of steroidal compounds 9a, 9b, 10a, 10b and their preparation from the commercially available 17α-acetoxyprogesterone.
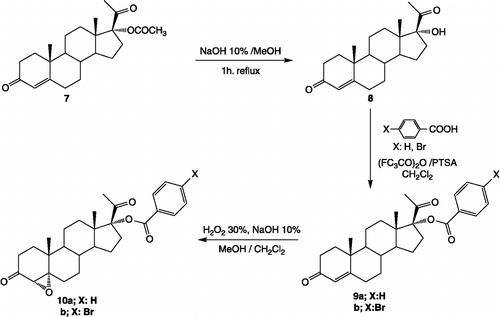
Table II. The IC50 values for finasteride and the synthesized steroids 9a, 9b, 10a, 10b with human prostate 5α–reductase enzyme. They represent the concentration of the steroid that inhibits 50% of 5α–reductase activity and were determined as described in the Experimental section.
![Figure 3. Inhibition of [3H] mibolerone binding to the androgen receptors by unlabeled steroids, as described in Methods section.](/cms/asset/959e1cb1-c52b-4794-81e2-c39f48d58e08/ienz_a_332539_f0003_b.gif)