Figures & data
Table I. Analytical data of divalent cobalt, nickel, copper, zinc and cadmium complexes derived from glyoxal and, 8-diaminonaphthalene.
Figure 3. Structure of the complexes. Where M = Co(II), Ni(II), Cu(II), Zn(II), Cd(II); X = Cl− 1, NO3− 1, CH3COO− 1.
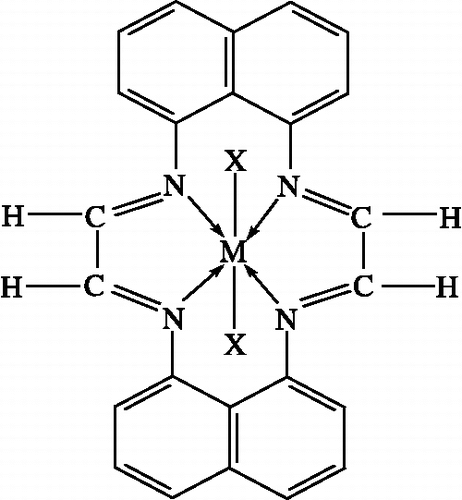
Table II. Minimum inhibitory concentration (MIC) of the complexes against test bacteria by using agar dilution assay.
Figure 4. Comparison of MIC of compounds with standard antibiotics up to the concentration 16 μg/mL. Bs—Bacillus subtilis (MTCC 8509); Bst—Bacillus stearothermophilus (MTCC 8508); Pp—Pseudomonas putida (MTCC 121); Ec—Escherichia coli (MTCC 51); Chloramphenicol, Streptomycin—standard antibiotics.
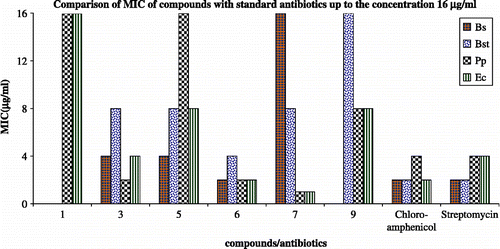
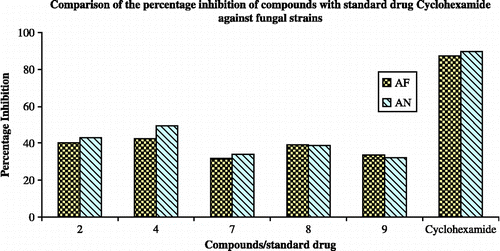
Table III. Antifungal (percentage inhibition) activities of the complexes against the fungal strains. (for concentration—100 μg/mL).
![Figure 1. 1H NMR spectrum of [Zn(C24H16N4)(OAc)2] complex.](/cms/asset/ee192ef0-cd9c-429d-be34-29d1d0622c67/ienz_a_339884_f0001_b.gif)
![Figure 2. FAB mass spectra of [Co(C24H16N4)(NO3)2] complex.](/cms/asset/dafebee0-d8de-4671-8151-9ca02851bf0c/ienz_a_339884_f0002_b.gif)