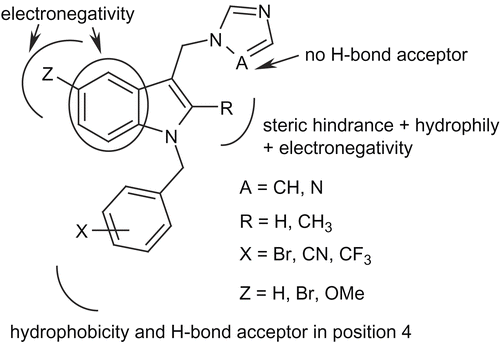
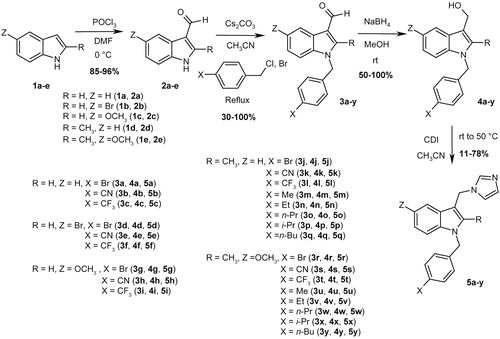
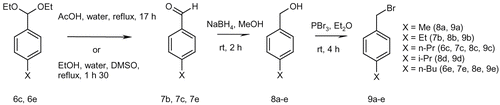
Figures & data
Table 1. Biological results of compounds 5a-l, 5r-t and comparison with predicted values of activity (boxes with * represent molecules used in the previous 3D-QSAR model).
Figure 3. Craig Diagram (σ: Hammett electronic substituent constant and π: Hansch hydrophobicity substituent constant).
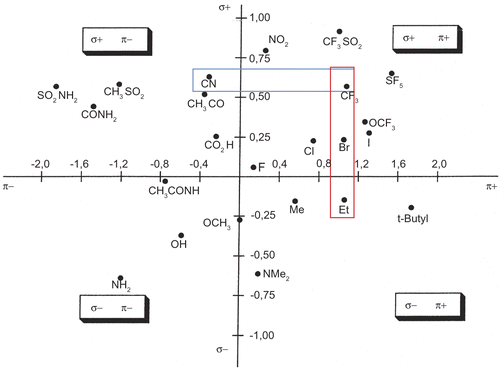
Table 2. Biological results of compounds 5m-q and 5u-y with alkyl side chains on para-position of the benzyl group.