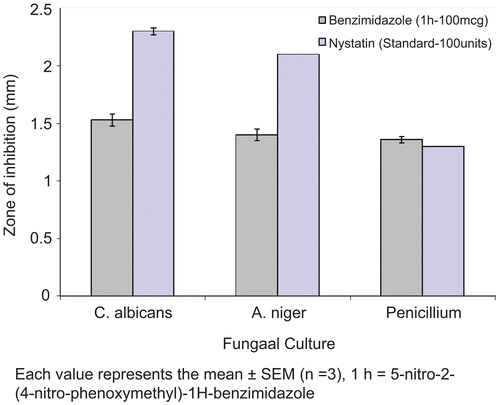
Figures & data
Table 1. Comparison between microwave-assisted and thermal method synthesis of 5-nitro-2-aryl substituted phenoxymethyl-1H-benzimidazole libraries.
Figure 1. Photographic comparison of compound 1h with standard for antibacterial activity in Staphylococcus aureus and antifungal activity in Penicillium.
A & B - Streptomycin (Standard) and Compound 1h for Staphylococcus aureus
C & D - Nystatin (Standard) and Compound 1h for Penicillium
A & C - c- control disc with DMSO, std- standard (Streptomycin-10mcg & Nystatin-100units)
B & D - a-1mcg, b-0.5mcg and c-0.25mcg
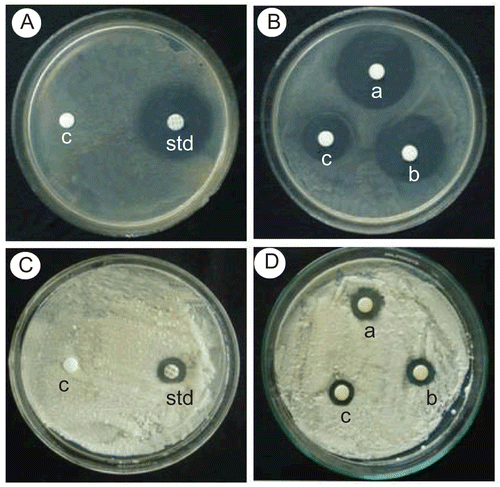
Figure 2. Bar curves for comparison of antibacterial activity of compound 1h with Streptomycin standard.
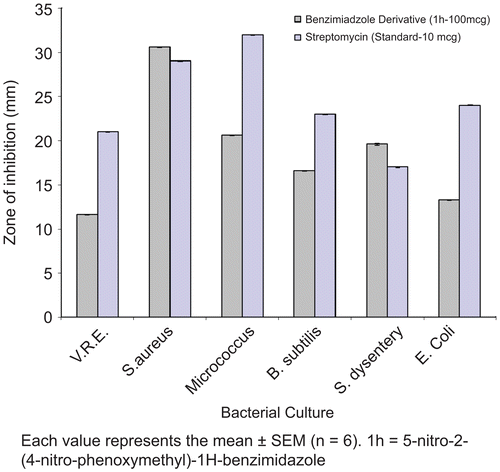
Table 2. The MIC values (μmoles/mL) of compounds 1a-1j for Antibacterial activity.
Table 3. The MIC values (μmoles/mL) of compounds 1a-1j Antifungal activity.