Figures & data
Table 1. Experimental data of the compounds 1–6.
Table 2. Spectral data of the compounds 1–6.
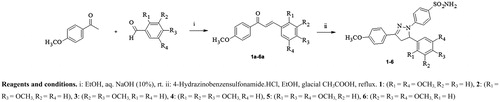
Scheme 1. Synthetic pathway of the pyrazoline bearing benzenesulfonamides 1–6. Reagents and conditions. i: EtOH, aq. NaOH (10%), rt. ii: 4-Hydrazinobenzensulfonamide.HCl, EtOH, glacial CH3COOH, reflux. 1: (R1 = R4 = OCH3, R2 = R3 = H), 2: (R1 = R3 = OCH3, R2 = R4 = H), 3: (R2 = R3 = OCH3, R1 = R4 = H), 4: (R1 = R2 = R3 = OCH3, R4 = H), 5: (R1 = R3 = R4 = OCH3, R2 = H), 6: (R2 = R3 = R4 = OCH3, R1 = H).