Figures & data
Table 1. The synthesis route of hetaryl sulfonamides 1–12.
Table 2. Physical and chemical characteristics of hetaryl sulfonamides 1–12.
Scheme 1. The synthesis routes of the 1-(4-methylsulfonyl)-2-thione-4-aryl-5-Z-6-methyl and oxyalkyl-imidazoles.
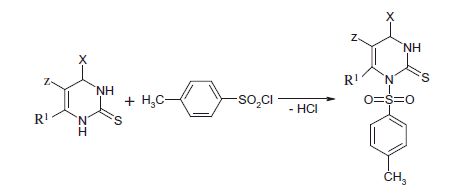
Table 3. The inhibition profiles of a dozen 1-(4-methylsulfonyl)-2-thione-4-aryl-5-Z-6-methyl and oxyalkyl-imidazoles 1–12 against human carbonic anhydrase isoenzymes I and II (hCA I and II), acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) enzymes.
Table 4. Determination of the half maximal concentrations (IC50, μg/mL) of Fe2+ chelating of several new hetaryl sulfonamides 1–12 and standard compounds including BHA, BHT, α-Tocopherol, Trolox and EDTA.
