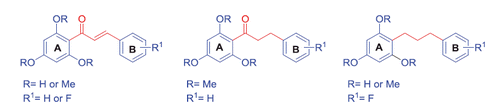
Figures & data
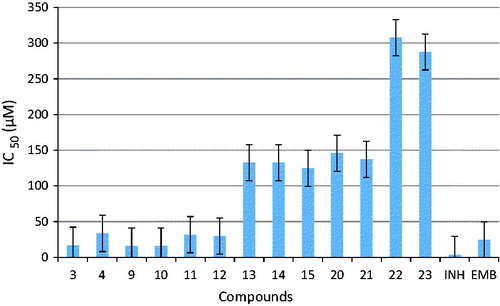
Figure 1. Comparative analysis of the antitubercular activity of the synthesized compounds and the standard drugs EMB and INH.
Scheme 2. Preparation of non- and fluoro-substituted trimethoxy/trihydroxy chalcones. Reagents and conditions: (i) 50% KOH, MeOH, (ii) H2, Pd-C, EtOH, (iii) 50% KOH, MeOH, (iv) H2, Pd-C, EtOH, (v) DIPEA, MOMCl, DCM, (vi) 50% KOH, MeOH, (vii) 12M HCl, EtOAc, (viii) H2, Pd-C, EtOH.
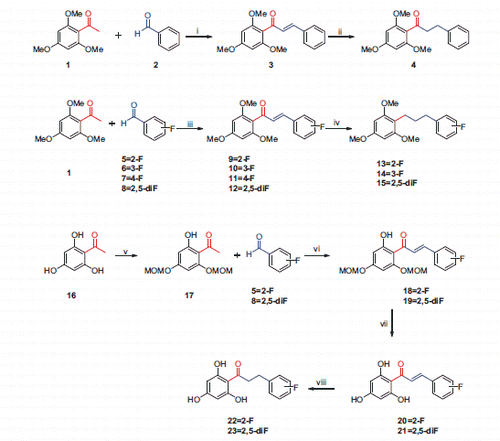
Table 1. Substitution patterns and antitubercular activity of target compounds (3, 4, 9–15, and 20–23).
Table 2. MICs of compounds 3, 4, 9–15, and 20–23 and the standard ampicillin against the selected bacterial strains.
Table 3. MICs of compounds 3, 4, 9–15, 20–23 and the standard fluconazole against selected fungi.