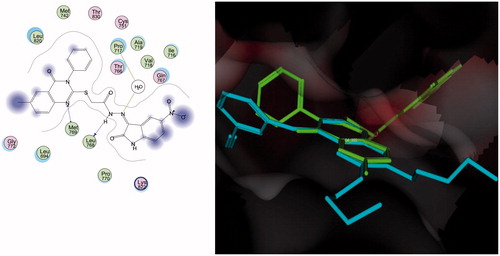
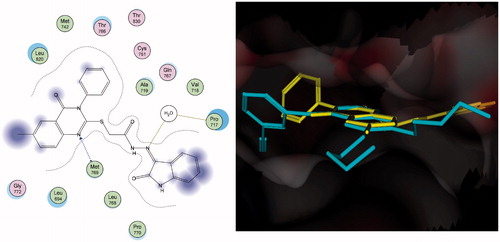
Figures & data
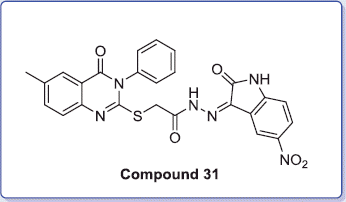
Figure 1. Reported and proposed quinazoline–isatin conjugates with antitumor and tyrosine kinase inhibitory activity.
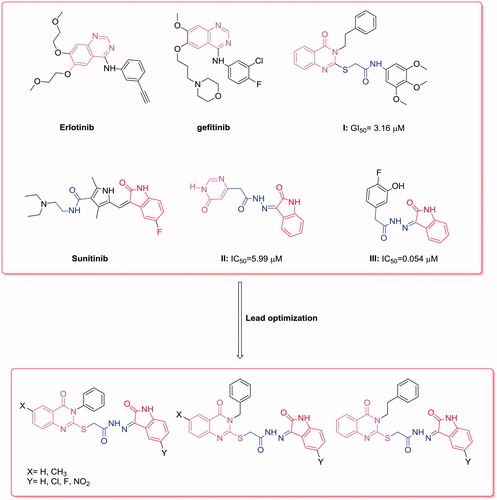
Table 1. In vitro antitumor activity of the newly synthesized compounds 16–34.
Figure 2. EGFR (left panel; green color) of MDA-MB-231 breast cell line and (right panel) MDA-MB-231 breast cell line after treatment with compound 31.
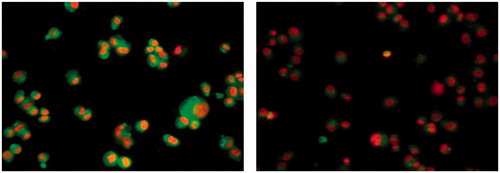
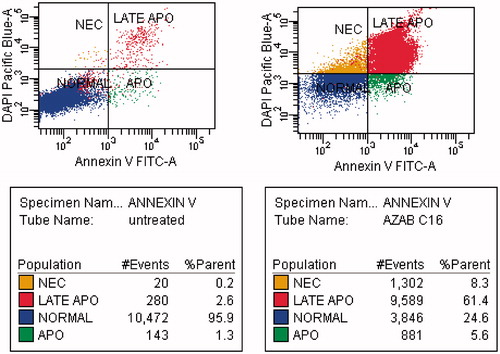
Figure 3. MDA-MB-231 breast cancer cell line was treated with compound 31 (right panel), which displayed an increased percentage of fluorescein isothiocyanate annexin V (Annexin V–FITC), and untreated control cells (left panel).

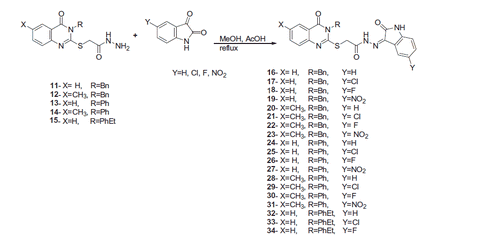
![Scheme 1. Synthesis of 2-[(3-substituted-4(3H)-quinazolinon-2-yl)thio]acetohydrazides 11–15.](/cms/asset/8ddff56f-ec1b-4523-9bd4-a8f5821713b1/ienz_a_1344981_sch0001.gif)