Figures & data
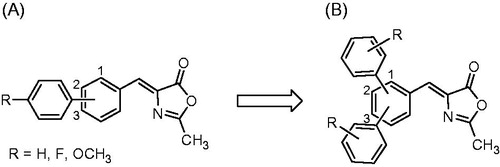
Figure 2. Structural evolution of methyleneoxazol-5(4H)-one scaffold: previously developed biphenyl 2-methyloxazol-5(4H)-one compounds (A) and newly synthesised terphenyl-2-methyloxazol-5(4H)-one derivatives (B).
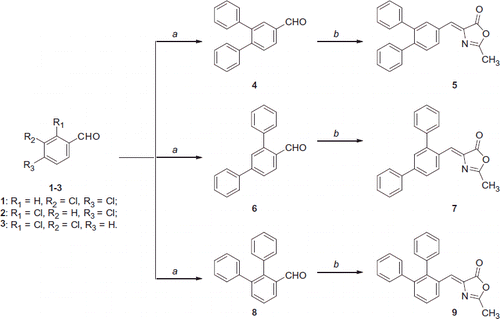
Scheme 1. Reagents and conditions: (a) for compound 4: phenylboronic acid, Pd(OAc)2, K3PO4, TBAB, H2O, 125 °C; for compound 6: phenylboronic acid, Pd(OAc)2, PPh3, aq. 2 M Na2CO3, toluene, EtOH, 100 °C; for compound 8: phenylboronic acid, Pd(OAc)2, PPh3, aq. 2 M Na2CO3, toluene, EtOH, 100 °C, then phenylboronic acid, Pd2(dba)3, Cs2CO3, Cy3P 20% toluene, dioxane, 100 °C; (b) N-acetylglycine, Ac2O, CH3COONa, reflux.
Scheme 2. Reagents and conditions: (a) phenylboronic acid, Pd(OAc)2, PPh3, aq. 2 M Na2CO3, toluene, EtOH, 100 °C; (b) N-acetylglycine, Ac2O, CH3COONa, reflux.
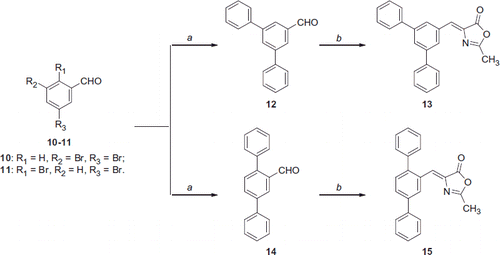
Scheme 3. Reagents and conditions: (a) phenylboronic acid, Pd2(dba)3, Cs2CO3, Cy3P 20% toluene, dioxane, 100 °C; b) N-acetylglycine, Ac2O, CH3COONa, reflux.
Scheme 4. Reagents and conditions: (a) variously substituted phenylboronic acid, Pd(OAc)2, PPh3, aq. 2 M Na2CO3, toluene, EtOH, 100 °C; (b) N-acetylglycine, Ac2O, CH3COONa, reflux.
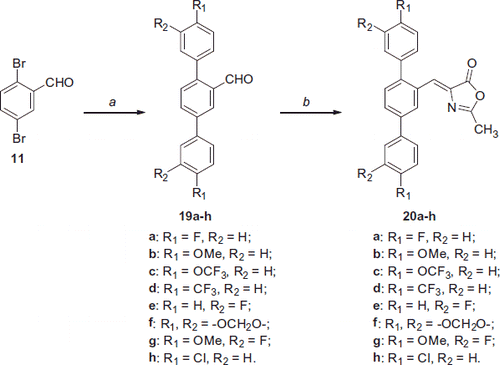
Table 1. Experimental inhibition activity (IC50) on human MAGL and FAAH of the analyzed compounds.
Figure 3. Compound 20b-MAGL inhibition analysis. (A) IC50 (µM) values of 20b at different preincubation times with hMAGL (0, 30 and 60 min). (B) Dilution assay: the first two columns indicate the inhibition percentage of compound 20b at a concentration of 10 and 0.25 µM. The third column indicates the inhibition percentage of compound 20b after dilution (final concentration = 0.25 µM).
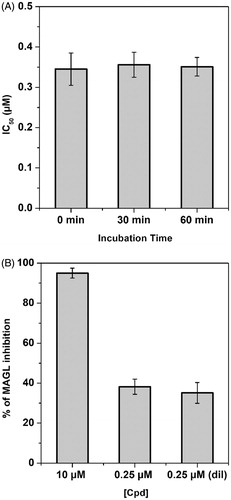
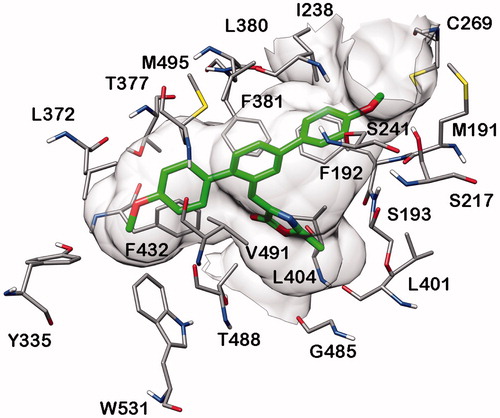
Figure 4. Minimised average structure of compound 20b docked into MAGL receptor (A) and analysis of 20b-MAGL H-bond interactions (B). The plot shows the distance analysis for the two H-bonds (i.e. HB1 and HB2).
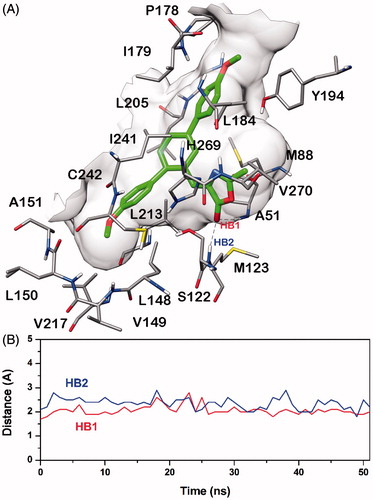
Table 2. MM-PBSA results for compound 20b docked into MAGL and FAAH.