Figures & data
Scheme 1. General synthetic route to 3-arylcoumarin, reagents, and conditions: (a) sulphur, p-toluenesulfonic acid, morpholine, 120 °C; (b) sodium hydroxide solution, tetrabutylammonium bromide, 100 °C; (c) acetic anhydride, triethylamine, 112 °C.
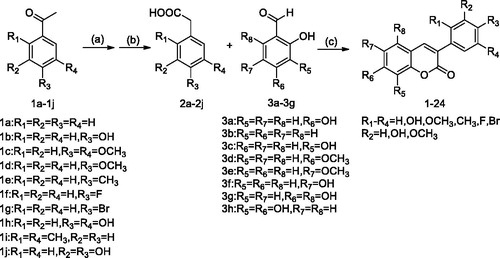
Table 1. Compounds 1–24.
Table 2. Biological evaluation in vitro.
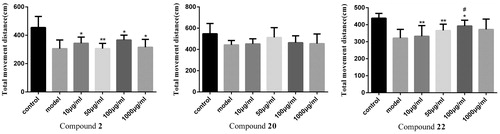
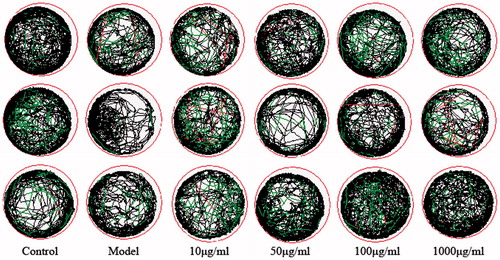
Figure 1. Effect of compounds 2, 20, 22 on the average total distance of zebrafish juveniles. (Compared with the control group, *p < .05 had a significant difference, **p < .01 had a very significant difference; compared with the model group, #p < .05 had a significant difference, ##p < .01 had a very significant difference).