Figures & data
Table 1. Activation of hCA isozymes I, II and BsuCA 1/FtuCA, with L-Trp, at 25 °C, for the CO2 hydration reactionCitation20.
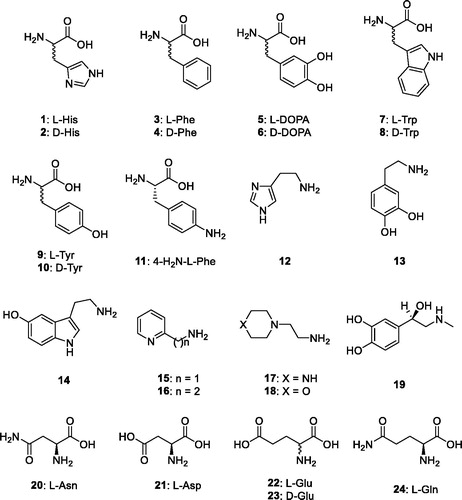
Table 2. Activation constants of hCA I, hCA II and the bacterial β-CAs investigated here with amino acids and amines 1–24, by a stopped-flow CO2hydrase assayCitation20.