Figures & data
Scheme 1. Syntheses of 1H-quinazolyl isoxazole-4-carboxamide derivatives. (i) EDC, HOBt, TEA, NH3 in MeOH, rt; (ii) BH3-THF, reflux; (iii) benzoyl chloride, CH2Cl2, 0 oC→ rt; (iv) (1) HCl/H2O/AcOH, μW, 150 oC, 10 min; (2) p-chloranil, toluene, reflux; (v) Fe, AcOH/H2O/EtOH, 60 oC; (vi) 5-methylisoxazole-4-carbonyl chloride, TEA, THF, rt.
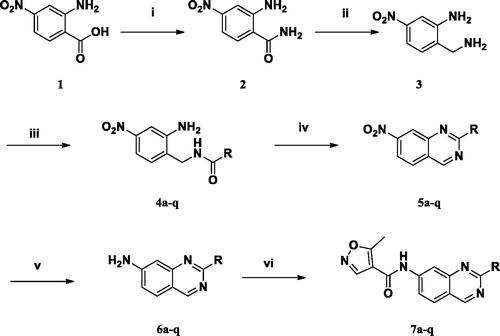
Table 1. Enzymatic activity of 5-methyl-N-(2-arylquinazolin-7-yl) isoxazole-4-carboxamide analogues.
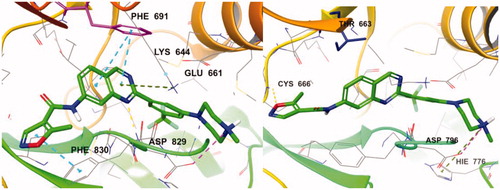
Figure 3. (Left) Compound 7d (green) at the active site of FLT3 (PDB: 4RT7); (right) 7e (yellow) at the active site of FLT3 (PDB: 4RT7).
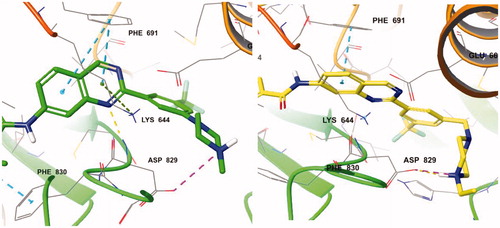
Figure 4. (Left) Compound 7n with equatorial O linkage (orange) at the active site of FLT3 (PDB: 4RT7); (right) compound 7n with axial O linkage (azure) at the active site of FLT3 (PDB: 4RT7).
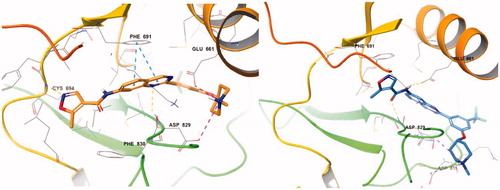
Table 2. Enzymatic activities of compound 7d against FLT3 mutants.


![Figure 5. Kinase profiling according to chemical scaffold. The profiles of the FMS kinase inhibitor [13] and quinazoline derivative 7d (10 μM) are shown.](/cms/asset/9b010760-9995-4cd8-97e3-0eec80929fa2/ienz_a_1758689_f0005_c.jpg)