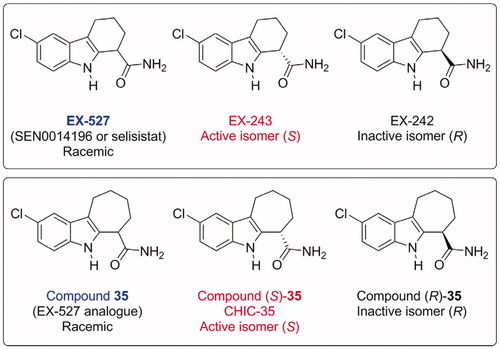
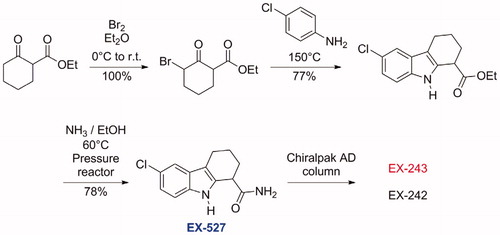
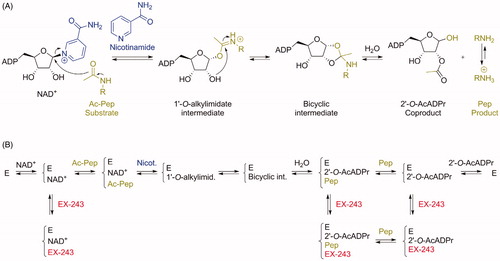
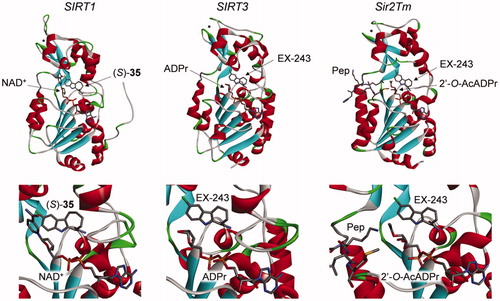
Figures & data
Table 1. In vitro assays of EX-527 and its analogue 35 on isolated recombinant sirtuins expressed in bacteria.
Table 2. Binding parameters of EX-527 with sirtuins.
Table 3. Representative examples of cellular effects of EX-527.
Table 4. Selected pharmacokinetics parameters of EX-527 (in plasma).
Table 5. Representative examples of in vivo assays of EX-527.