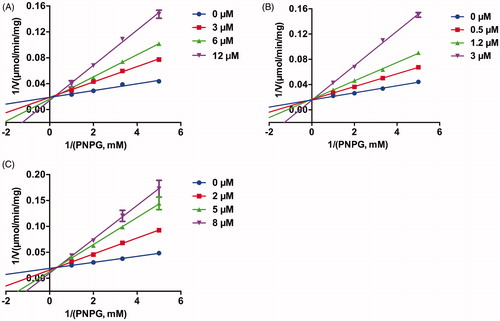
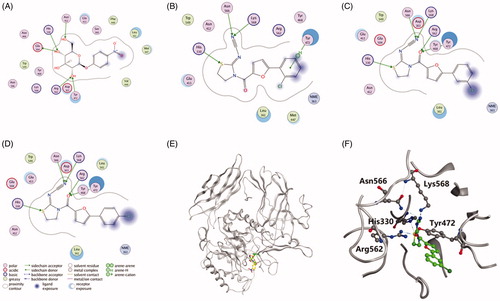
Figures & data
Figure 1. General synthetic procedure for title compounds 1–13. R = 1: 2-Cl; 2: 3-Cl; 3: 4-Cl; 4: 2-F; 5: 4-F; 6: 2,4-di-F; 7: 2,6-di-F; 8: 2-NO2; 9: 4-NO2; 10: 4-Br; 11: 4-Me; 12: 4-OMe; 13: H.
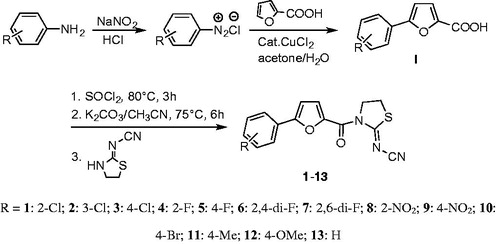
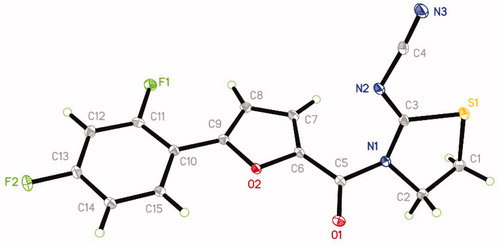
Table 1. Chemical structures of 13 thiazolidin-2-cyanamide derivatives and the inhibitory activity against EcGUS-mediated PNPG hydrolysis.
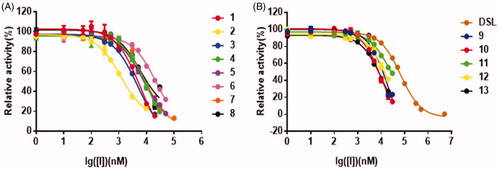
Figure 3. The dose-dependent inhibition curves of inhibitors on PNPG-hydrolyzing activity of EcGUS. (A) Compounds 1–8; (B) compounds 9–13, and DSL. All data were expressed as mean ± standard deviation of triplicate reactions.