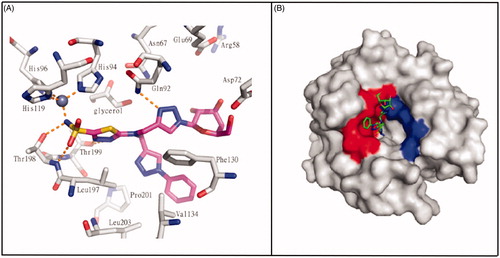
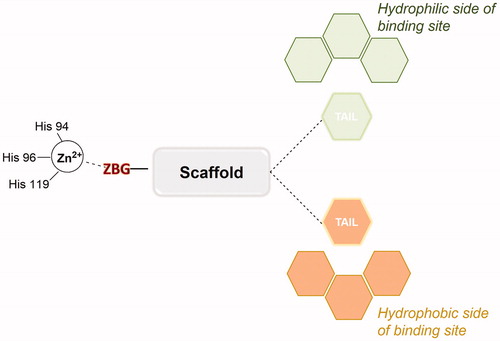
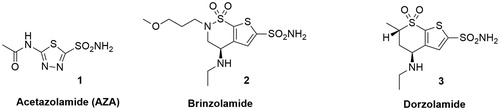
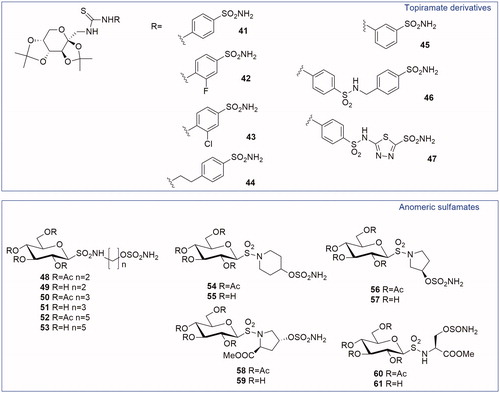
Figures & data
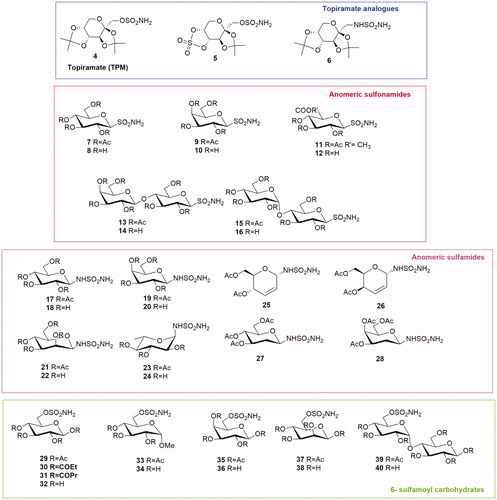
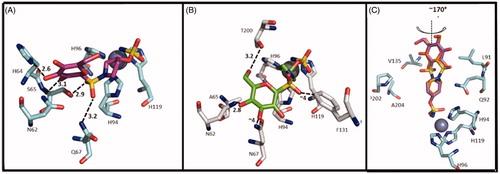
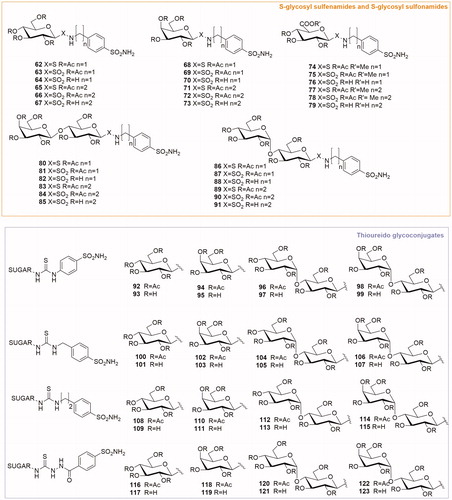
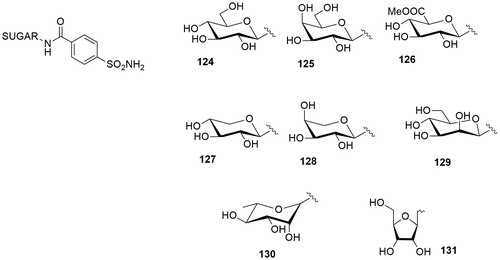
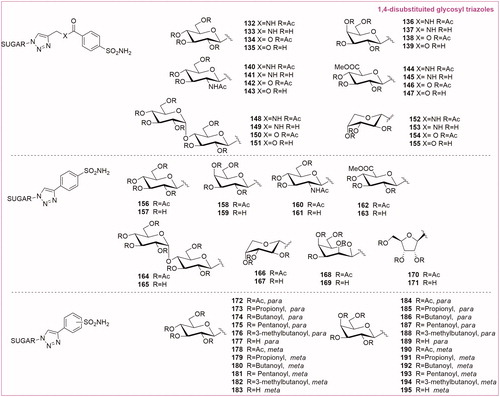
Table 1. Chemical structure, CA IX inhibitory activity, CA I/ CA IX selectivity profile of the best compound for each class of CA glycosidic inhibitors
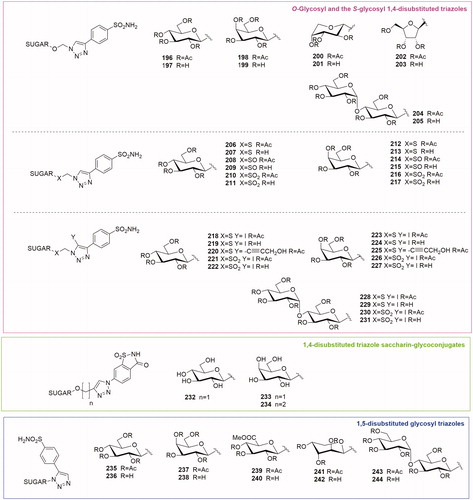
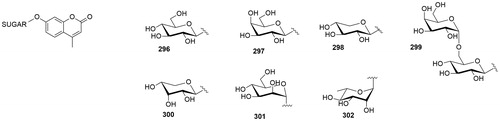
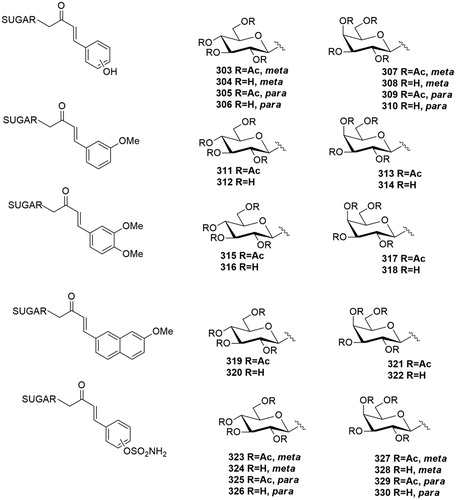
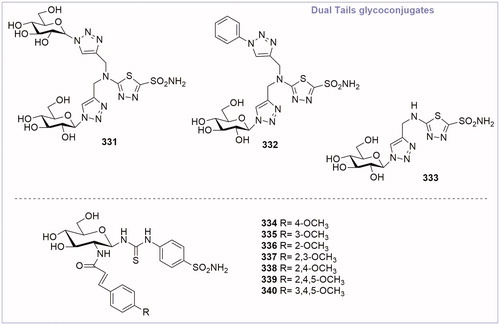
Table 2. Chemical structure, CA IX inhibitory activity, CA I/ CA IX selectivity profile of the best compound for each class of CA glycoconjugated inhibitors