Figures & data
Figure 1. General structure of taurine sulphonamides terminated with thioureido tailsCitation15.
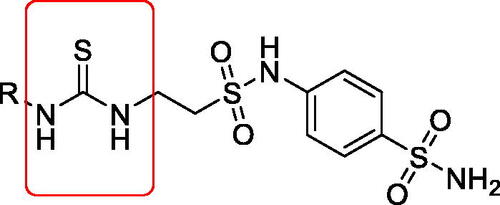
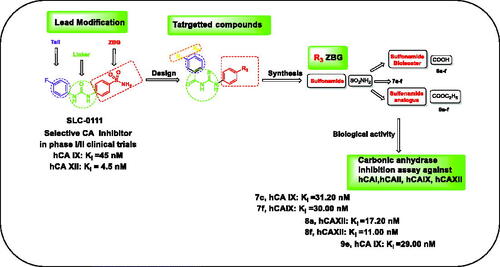
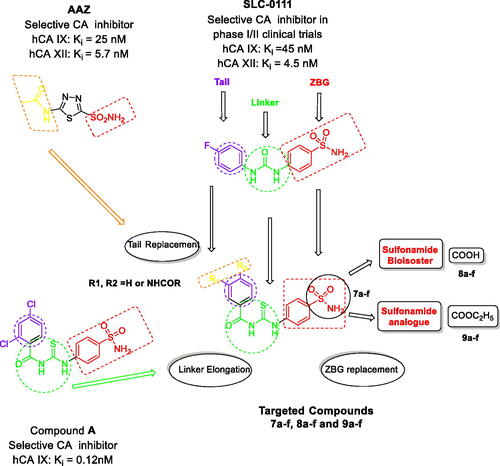
Figure 2. The design for the targeted compounds benzoylthioureido benzenesulfonamide derivatives (7a–f) and their analogues (8a–f) and (9a–f).
Scheme 1. Synthetic pathways for compounds 7a–f, 8a–f, 9a–f. Reagents and conditions: (i) KOH, acetonitrile, R.T, 1–2 h, (ii) SOCl2, methylene chloride, reflux, 4–5 h, (iii) NH4SCN, acetone, reflux, 1–3 h, (iv) acetone, reflux, 2–3 h.
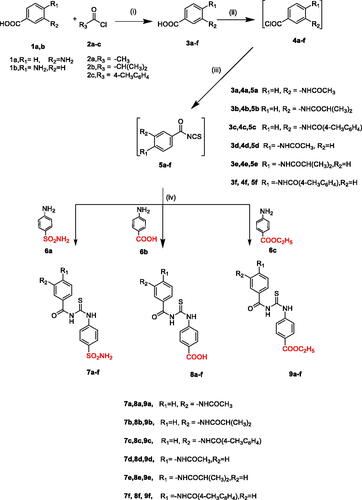
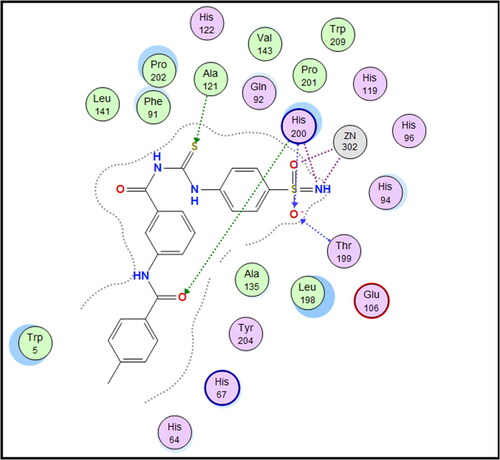
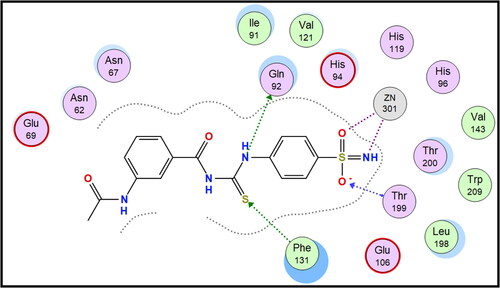
Table 1. Carbonic anhydrase inhibitory activity of compounds 7a–f, 8a–f, and 9a–f and the standard sulphonamide inhibitor acetazolamide (AAZ) using a stopped flow CO2 hydrase assay.
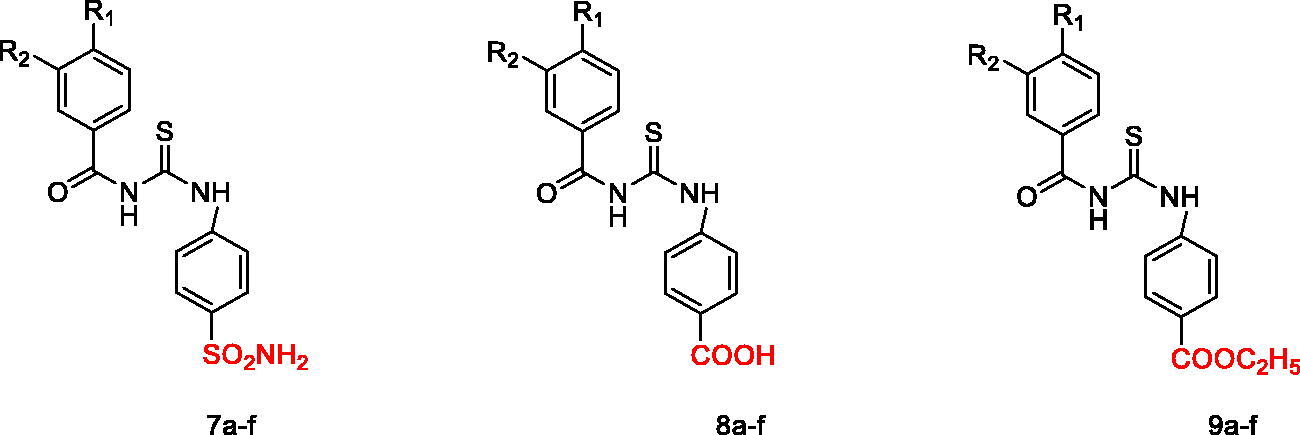
Table 2. Calculated selectivity indexes (S.I.s) for inhibition of hCA IX over hCA I and hCA II isoforms for compounds 7c, 7f, 9e, SLC-0111, and AAZ.
Table 3. Calculated selectivity indexes (S.I.s) for inhibition of hCA XII over hCA I and hCA II isoforms for compounds 8a, 8f, SLC-0111, and AAZ.
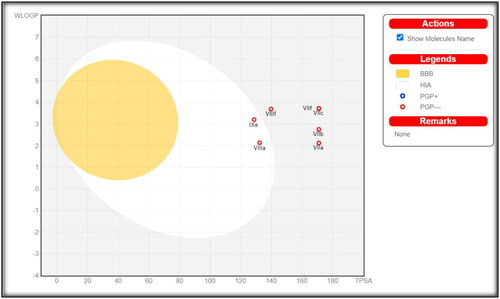
Figure 9. Expected boiled-egg plot from Swiss ADME online web tool for compounds 7a, 7b, 7c, 7f, 8a, 8f, and 9e.