Figures & data
Table 1. PBP1b inhibitory activity and antimicrobial effect (MIC) of boronic acid derivatives.
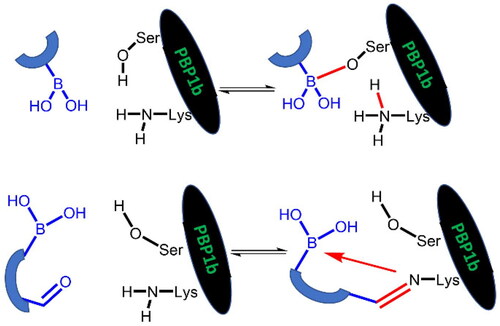
Scheme 2. (A) Complex formation of penicillins with penicillin-binding protein. Bulky groups near the covalent enzyme-ligand bond are encircled. (B) Complex formation of branched boronic acids with penicillin-binding proteins.
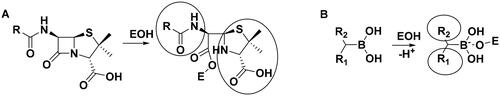
Table 2. PBP1b inhibitory activity and antimicrobial effect (MIC) of branched boronic acids.
Scheme 6. Serine activation and attack on the boronic acid. Electron movements are shown by curly arrows. Interatomic distances used for defining reaction coordinates are shown with straight arrows.
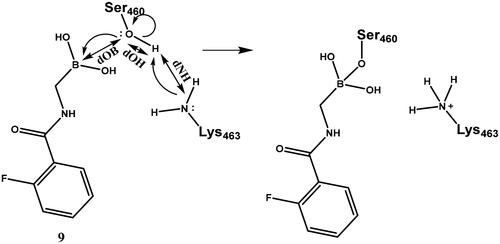
Table 3. Experimental activities, and computed barrier and reaction free energies for selected boronic acids towards PBP1b.
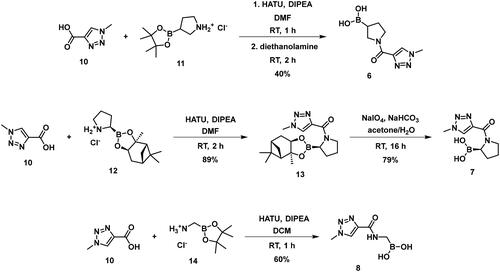
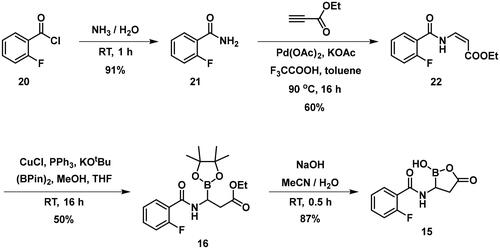
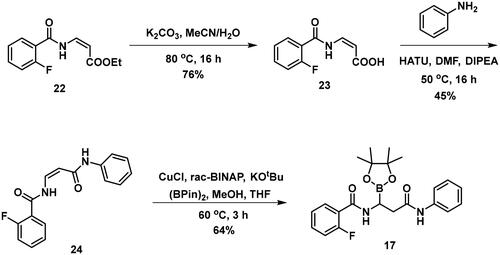
Table 4. PBP1b inhibitory activity and antimicrobial effect (MIC) of potential lysine labelling boronic acid derivatives.