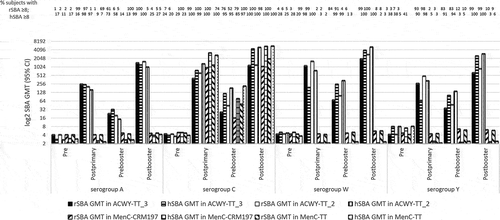
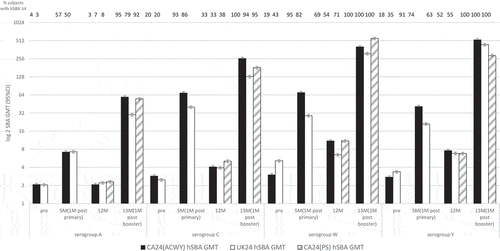
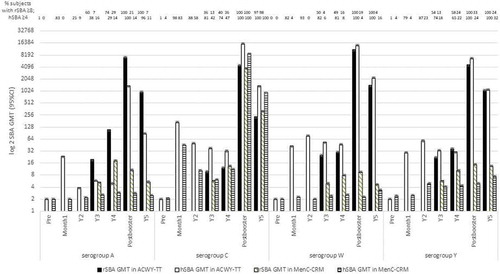
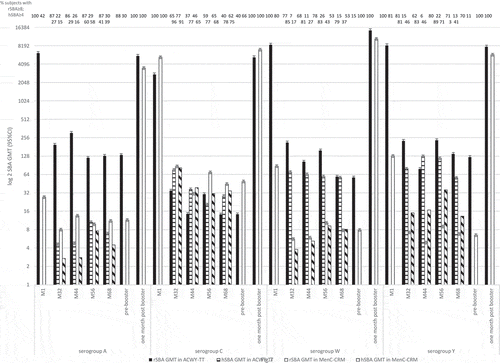
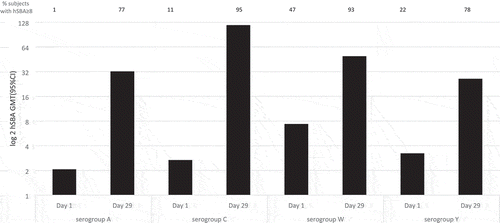
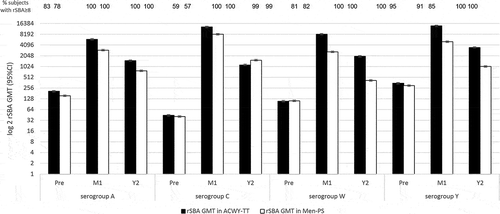
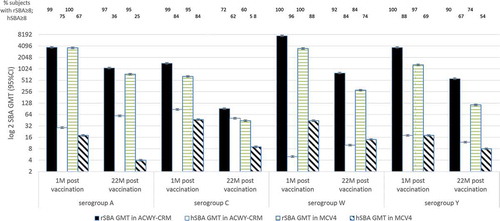
Figures & data
Merino Arribas JM, Carmona Martínez A, Horn M, et al. Safety and immunogenicity of the quadrivalent meningococcal serogroups A, C, W and Y tetanus toxoid conjugate vaccine coadministered with routine childhood vaccines in European infants. Pediatr Infect Dis J. 2017;36(4):e98–e107. Perrett K, Snape M, Ford K, et al. Immunogenicity and immune memory of a nonadjuvanted quadrivalent meningococcal glycoconjugate vaccine in infants. Pediatr Infect Dis J. 2009;28(3):186–193. Vesikari T, Forsten A, Bianco V, et al. Antibody persistence up to 5 years after vaccination of toddlers and children between 12 months and 10 years of age with a quadrivalent meningococcal ACWY-tetanus toxoid conjugate vaccine. Hum Vaccin Immunother. 2015;12(1):132–139. Knuf M, Romain O, Kindler K, et al. Immunogenicity and safety of the quadrivalent meningococcal serogroups A, C, W-135 and Y tetanus toxoid conjugate vaccine (MenACWY-TT) in 2–10-year-old children: results of an open, randomised, controlled study. Eur J Pediatr. 2013;12(5):601–612. Knuf M, Helm K, Kolhe D, et al. Antibody persistence and booster response 68 months after vaccination at 2–10 years of age with one dose of MenACWY-TT conjugate vaccine. Vaccine. 2018;36(23):3286–3295. Huang L, Chiu N, Yeh S, et al. Immunogenicity and safety of a single dose of a CRM-conjugated meningococcal ACWY vaccine in children and adolescents aged 2–18 years in Taiwan: results of an open label study. Vaccine. 2014;32(40):5177–5184. Quiambao B, Jain H, Bavdekar A, et al. Persistence of the immune response two years after vaccination with quadrivalent meningococcal ACWY-tetanus toxoid conjugate vaccine (MenACWY-TT) in Asian adolescents. Hum Vaccin Immunother. 2016;12(8):2162–2168. Gill C, Baxter R, Anemona A, et al. Persistence of immune responses after a single dose of Novartis meningococcal serogroup A, C, W-135 and Y CRM-197 conjugate vaccine (Menveo®) or Menactra® among healthy adolescents. Hum Vaccin. 2010;6(11):881–887.