Figures & data
Table 1. Hexaxim composition (per 0.5 mL dose).
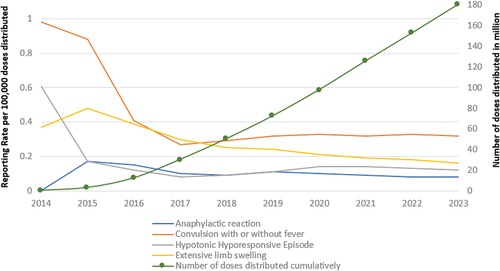
Figure 1. Hexaxim use in clinical practices around the world in the past decade (2013 – 2023).
Figure 2. Seroprotection/Seroconversion rates 1 month after a primary vaccination with 2 or 3 doses of Hexaxim.
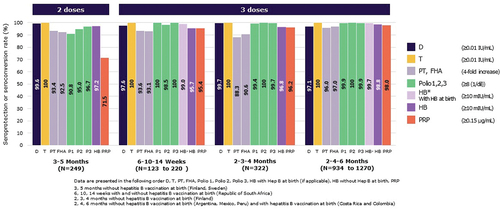
Figure 3. Seroprotection/Seroconversion rates 1 month after a booster vaccination of Hexaxim in the second year of life.
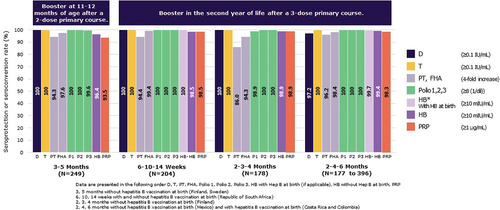
Table 2. Summary of Hexaxim clinical trials (primary series, booster, and antibody persistence).
Figure 4. Persistent immune memory to HB following 2, 4, 6 month primary series vaccination of Hexaxim and no booster.
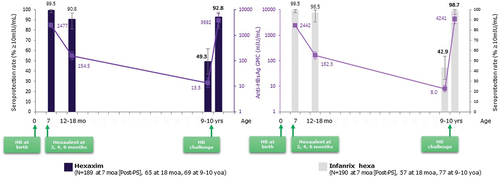
Table 3. Solicited reactions after any infant vaccination – integrated analysis of Hexaxim versus Infanrix hexa.
Table 4. Characteristics of AE reports and the 10 most common events reported after DTaP-IPV-HB-Hib vaccination (June 1, 2013 to April 17, 2023).
Table 5. Observed versus expected analysis for Sudden Infant Death Syndrome/Sudden Death in the European Economic Area.