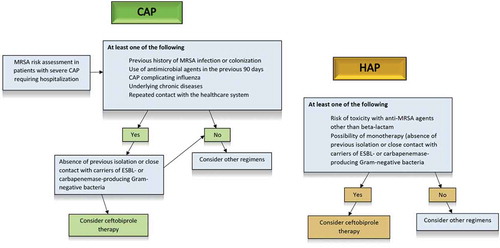
Figures & data
Table 1. Ceftobiprole activity against clinical isolates of various bacterial species, from large surveillance studies.
Table 2. Pharmacokinetic (PK) parameters of ceftobiprole after single 500 mg intravenous dose over 2 h infusion in healthy volunteers [Citation25,Citation29].
Table 3. Efficacy data from phase 3, non-inferiority, randomized clinical trials in patients with CAP and HAP/VAP.