Figures & data
Figure 1 Patient disposition from enrollment to eligibility. ARV, antiretroviral; ATV/r, ritonavir-boosted atazanavir; NRTI, nucleos(t)ide reverse transcriptase inhibitors.
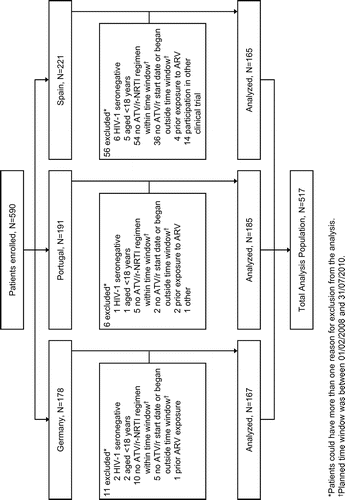
Table 1 Demographic and baseline characteristics
Figure 2 Time to discontinuation of treatment (A) and time to virologic failure using ≥ 50 copies/mL cut-off (B) and using ≥ 500 copies/mL cut-off (C). ATV/r, ritonavir-boosted atazanavir; BL, baseline; CI, confidence interval.
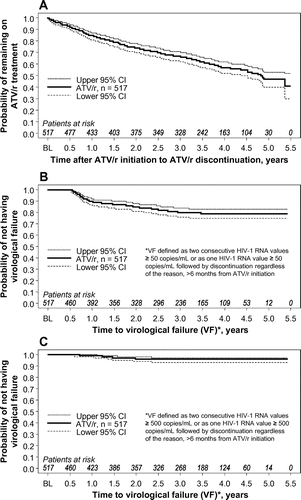
Table 2 Univariate and multivariate Cox proportional hazards models for time to discontinuation of an ATV/r-based regimen
Table 3 Reasons given by the investigator for discontinuation
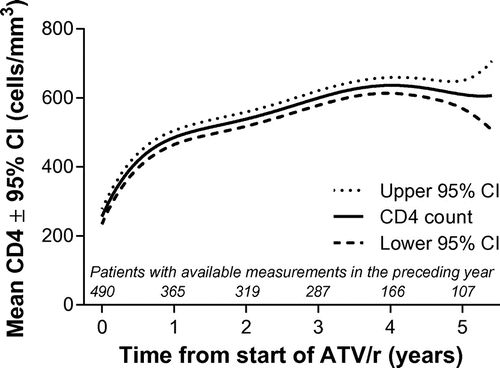
Figure 3 Mixed-model repeated measures estimates of mean CD4 count over time. ATV/r, ritonavir-boosted atazanavir; CI, confidence interval.