Figures & data
Table 1 Baseline characteristics of patients analyzed by proviral DNA genotyping (N = 51).
Table 2 Protease and reverse transcriptase drug resistance mutations detected by proviral DNA and historical RNA genotypes at baseline
Table 3 Week 48 virologic outcomes of patients with pre-existing NRTI or NNRTI resistance mutations by baseline proviral DNA or historical RNA genotype
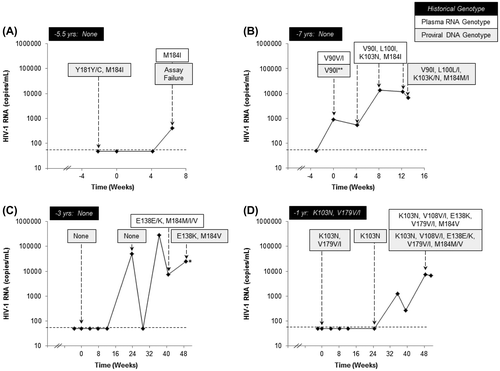
Figure 1 (A–D) HIV-1 RNA (copies/mL) curves and genotyping results for the four RPV/FTC/TDF-treated patients who experienced virologic failure with emergent resistance. Historical genotype results and timing prior to study enrollment are shown in black boxes. Plasma RNA genotype results from visits on study drug are indicated in white boxes. Proviral DNA genotype results from PBMCs collected during the study are denoted in gray boxes. The baseline visit is indicated as Week 0 with the screening time point occurring within 4 weeks prior. (*) sample was collected after the patient discontinued study drug; (**) result was obtained using an earlier version of the proviral DNA assay that used population sequencing.