Figures & data
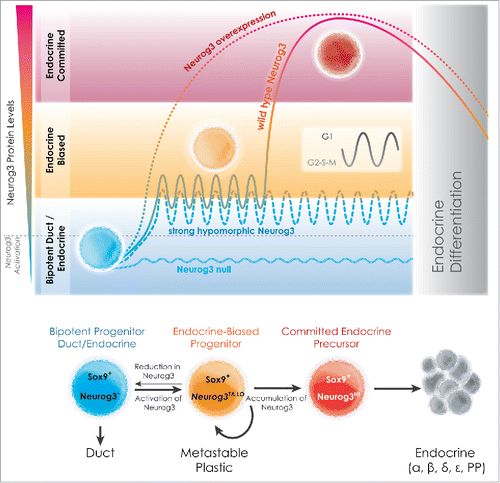
Figure 1. Endocrine-biasing and cell-cycle regulation - a model for lineage-priming in pancreas organogenesis. Normally, low-level Neurog3 activation in Sox9+ progenitors defines an actively cycling endocrine-biased progenitor state (orange). Neurog3 levels are postulated to oscillate over the cell cycle (inset), with G1 lengthening promoting Neurog3 accumulation, and its targeting of genes that cause endocrine commitment. While increasing (by Neurog3 overexpression) or eliminating Neurog3 expression (Neurog3 null) enforces or blocks endocrine-lineage allocation, respectively, greatly reducing Neurog3 (strong hypomorph) perpetuates the mitotic state, and allows drift from this metastable state to other lineages. Figure courtesy of Jean-Philippe Cartailler.