Figures & data
Figure 1. Architecture of an epithelial cell in interphase and entering mitosis. (a) representation of a cell in interphase in a pseudostratified epithelum. Cells are oriented along the apical-based axis. The apical pole (top) is exposed to the lumen while the basal pole (bottom) is in contact with the basement membrane, notably through integrins. Polarity complexes ensure the establishment and maintenance of polarity and cell-cell adhesion. (b) cell in prophase/prometaphase. When cells enter mitosis, actin cytoskeleton reorganization and osmotic swelling lead to cell rounding, which generates pushing forces towards the environment. Adherences (unctionsCell-cell, as well as cell-matrix, adhesions are transiently downregulated
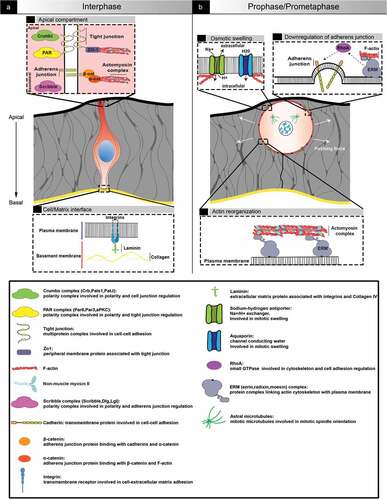
Figure 2. Examples of non-apical mitosis occurrence in development. (a) Representation of a transverse section from a gastrulating embryonic day (E7) mouse embryo. At the primitive streak (right square), cells undergo epithelial to mesenchymal transition and migrate away from the epibast. Mitosis can be found all along the apical-basal axis. (b) Representation of a neural tube transverse section from an HH13+ (-48h) chick embryo. Neural crest cells delaminate from the neural tube roof plate, and mitosis can be found all along the apical-basal axis. (c) Representation of interkinetic nuclear migration. In pseudostratified epithelia, nuclei are classically translocated during G2 and cells undergo mitosis apically
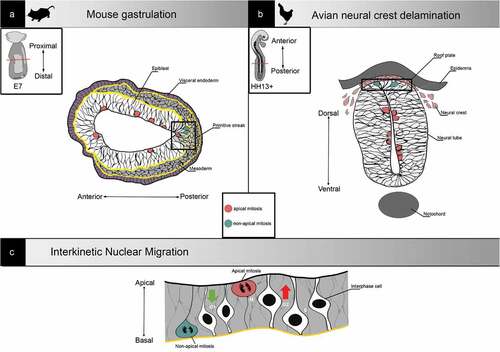
Figure 3. Examples of tissue reorganisation associated with mitosis in development. (a) In chick, during gastrulation, mitosis is required for spatial patterning by favouring cell rearrangement and tissue fluidity. (b) In fly, during trachea invagination, mitosis accelerates the process via the generation of pushing forces against the environment. (C) In zebrafish, during dome formation, mitosis supports tissue phase transition by interfering with cell-cell adhesion
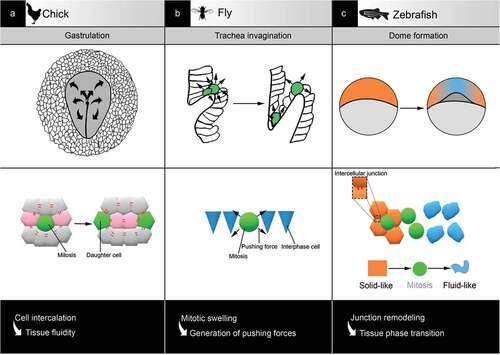
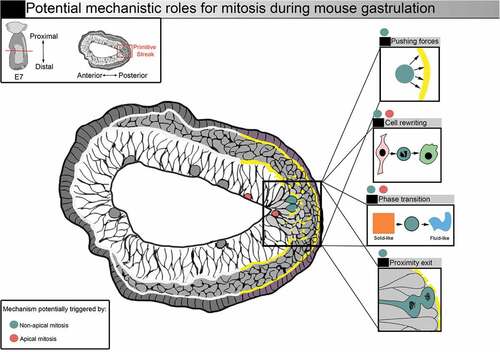
Figure 4. Potential mechanistic roles for mitosis during mouse gastrulation. Representation of a transverse section from a E7 mouse embryo. Squares on the right illustrate potential mechanistic roles for mitosis. Increased mitotic index (red and blue dots) could facilitate nascent mesoderm cell exit. Mitosis could benefit gastrulation by supporting shape and fate transition via mitotic cell rewriting. Forces generated by mitotic cells, as well as the remodelling of cell-cell adhesions, could also favour tissue phase transition. Entering mitosis in basal position (blue dots) could favor basement membrane breakage, and the generation of daughter cells close to the exit could facilitate delamination