Figures & data
Table 1. Mode of action of the insulin/IGF ligands.
Table 2. Effects of constitutive gene KO for the insulin/IGF-receptors signal components on body/organ size.
Table 3. Summary of insulin/IGF signal transducers acting as oncogenic factors versus tumor suppressors.
Table 4. Downstream signal targets and relevant PTM sites for the insulin/IGF signal transduction components implicated in cell growth and cell-cycle control.(a) www.phosphositeplus.com DB, (b) UniProt, [Citation301].

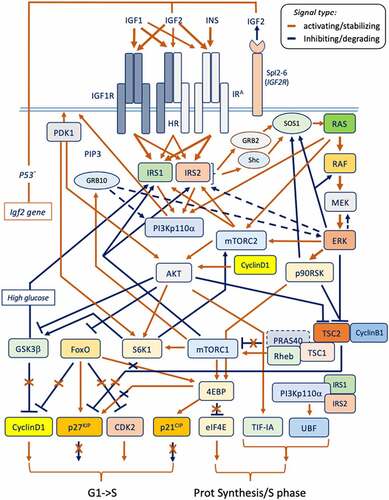
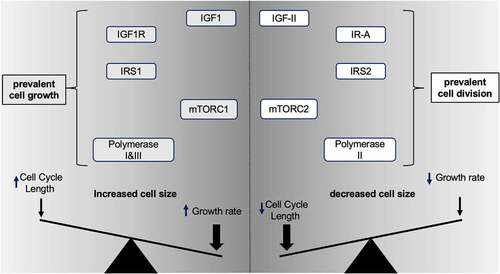
.](/cms/asset/88703da2-84d2-4c40-89ae-7de3e72abef7/kccy_a_2108117_f0004_b.gif)
