Figures & data
Figure 1 Human Pulmonary Dendritic cells. Pulmonary dendritic cells (DCs) are identified by flow cytometry within the cell population that is low autofluorescent (to distinguish them from macrophages which are high autofluorescent), HLA-DR +ve and that does not express lineage markers (CD3, CD14, CD16, CD19, CD20 and CD56). Within this cell population, different markers identify plasmacytoid (pDC) and different myeloid DC (mDC) subsets. mDC and pDC express different types of Toll-like receptors (TLR). Mature DCs are identified by upregulation of specific maturation markers such as co-stimulatory molecules and the chemokine receptor CCR7. (BDCA: Blood Dendritic Cell Antigen; DC-SIGN: Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin; MHC: Major Histocompability Complex; DC-LAMP: Dendritic Cell Lysosome-Associated Membrane glycoprotein). (*): BDCA-3 is also expressed on pDCs in broncho-alveolar lavage fluid.
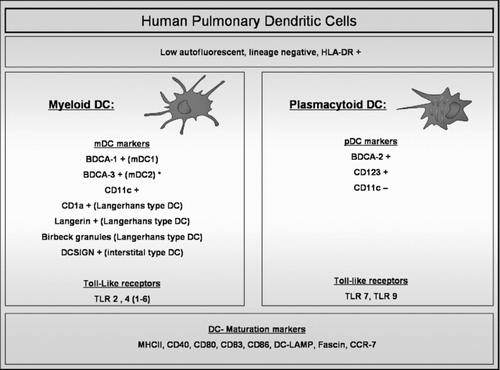
Table 1 Studies of Pulmonary Dendritic cells in Smokers and COPD patients
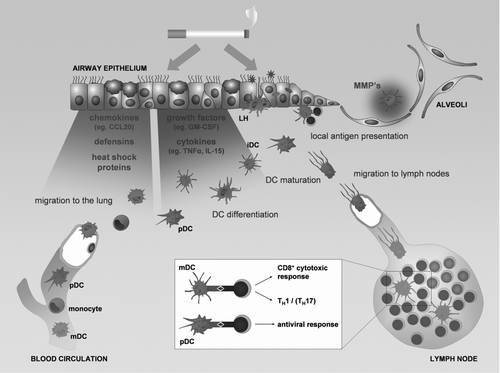
Figure 2 The role of dendritic cells in the pathogenesis of COPD. Chemokines, defensins and heath shock proteins produced by the lung tissue in response to the damaging effect of cigarette smoke attract circulating dendritic cells precursors (blood myeloid dendritic cells (mDC), plasmacytoid dendritic cells (pDC) and monocytes). The differentiation of the DC precursors into different subsets will be modulated by the growth factors and cytokines released by the damaged lung tissue, resulting in an altered DC population in the lung with increased numbers of Langerhans type DCs (LH). Together with the interstitial type DCs (iDC) and the pDCs, they sample antigens and sense for danger signals in the mucosal area. Maturing DCs migrate towards the lymph nodes where they present antigens to lymphocytes and orchestrate the outcome of the adaptive immune response. DCs can also present their antigens locally in the airway mucosa. The process of maturation can be directly attenuated by cigarette smoke. The altered DC population can skew the adaptive immune response towards a T helper 1, CD8+ cytotoxic T cell or T helper 17 profile. In addition DCs can contribute to the protease-antiprotease imbalance by producing metalloproteinases (MMP's), involved in the development of emphysema.