Figures & data
Figure 1 Immunocytochemistry of cultured cell lines engineered to ectopically express untagged Panx1 or Panx3. The localization of Panx1 (A, C, E, G, and I; red) and Panx3 (B, D, F, H and J; red) were revealed by immunolabeling with purified anti-Panx1 and anti-Panx3 antibodies, respectively. Inserts in A and B depict peptide pre-adsorption assays for each pannexin antibody. Arrowheads denote different pannexin cell surface distribution profiles in NRK (A) and REKs (C, D); Panx1 localization in a perinuclear compartment (E) and Panx3 at the cell surface (F). Nuclei are depicted in blue with Hoechst 33342 (A–D; G–J). Bars = 10 μ m
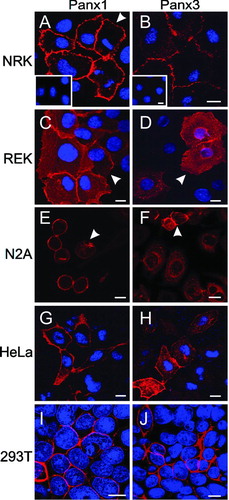
Figure 2 Subcellular expression profiles of N-glycosylation mutants Panx1N254Q and Panx3N71Q. Mutant pannexins were expressed in NRK (A, B), 293T (C, D), and HeLa (E, F) cells prior to immunolabeling for Panx1 (A, C, E; red) or Panx3 (B, D, F; red). Note the presence of both mutants at the cell surface as well as within intracellular compartments. Nuclei were stained with Hoechst 33342 (blue). Bars = 10 μ m.
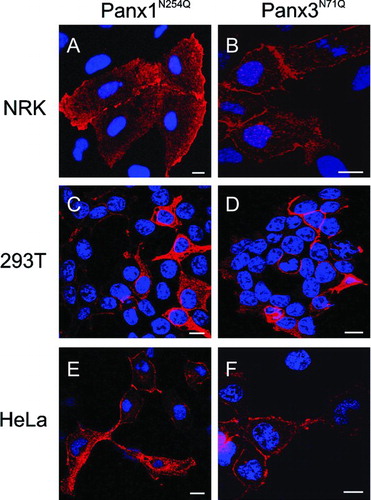
TABLE 1 Incidences of dye transfer in pannexin and mutant expressing HeLa cells
Figure 3 Expression of endogenous Panx1 and Panx3 in cultured cell lines and primary osteoblasts. Immunolabeling of the mouse osteoblast cell line MC3T3-E1 (A, C, E, G; red) for Panx1 or Panx3, or primary osteoblasts for Panx1 (A, insert) revealed that these pannexins were localized to intracellular compartments. Immunolabeling of MDCK cells (B, D, F, H) for Panx1 or Panx3 revealed that these pannexins were evenly distributed at the cell surface in most cells while some cells exhibited a cell surface stitching pattern (B, F, inserts). When the antibodies were premixed with the cognate peptides used to generate the anti-pannexin antibodies, the vast majority of labeling was eliminated (C, D, G, H). Nuclei were stained with Hoechst 33342 (A, C, E, G). Bars = 10 μ m.
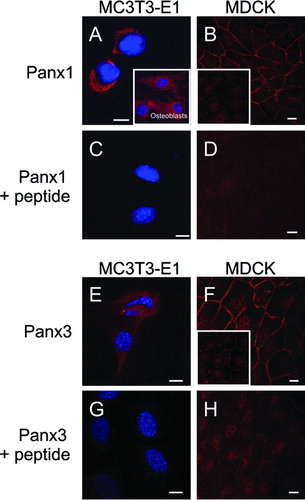
TABLE 2 Summary of pannexin 1 and pannexin 3 characteristics