Figures & data
Figure 1. Classification of 20 common amino acids considering their volume, hydropathy, and chemical classes as well as their charge and polarity [Citation11].
![Figure 1. Classification of 20 common amino acids considering their volume, hydropathy, and chemical classes as well as their charge and polarity [Citation11].](/cms/asset/71e7ea68-733d-4bfa-9957-a7cd3b539df4/lspr_a_2038625_f0001_oc.jpg)
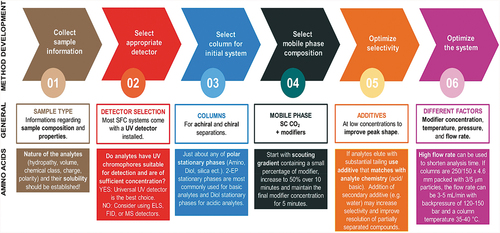
Figure 2. Flow scheme of laboratory-scale SFC: V1-V3 – valves, AV – automated valve, PG – pressure gauge [Citation57].
![Figure 2. Flow scheme of laboratory-scale SFC: V1-V3 – valves, AV – automated valve, PG – pressure gauge [Citation57].](/cms/asset/0bdf0659-80b9-4e25-8f46-b43f1eb437f8/lspr_a_2038625_f0002_b.gif)
Figure 3. Flow scheme of pilot preparative to production scale SFC: 1 - CO2 tank, 2 - CO2 cooler, 3 - CO2 pump, 4 - CO2 heater, 5 - modifier tank, 6 - modifier pump, 7 - column with piston, 8 - sample tank, 9 - detector, 10 - flow meter, 11 – evaporator [Citation76].
![Figure 3. Flow scheme of pilot preparative to production scale SFC: 1 - CO2 tank, 2 - CO2 cooler, 3 - CO2 pump, 4 - CO2 heater, 5 - modifier tank, 6 - modifier pump, 7 - column with piston, 8 - sample tank, 9 - detector, 10 - flow meter, 11 – evaporator [Citation76].](/cms/asset/9a85b4d6-a25c-4884-a23a-bb6f01018893/lspr_a_2038625_f0003_b.gif)
Figure 4. Specific mobile and stationary phases in SFC selected according to the polarity of soluble substances [Citation54].
![Figure 4. Specific mobile and stationary phases in SFC selected according to the polarity of soluble substances [Citation54].](/cms/asset/0859a185-e00a-4206-9ec4-7509b90b68e9/lspr_a_2038625_f0004_oc.jpg)
Table 1. Chiral separations of amino acids and amino acid derivatives with SFC using different stationary and mobile phases
Figure 6. Comparison of chromatograms of FMOC-DL-Alanine-OH in RP-HPLC and SFC. Experimental conditions: column: Lux™ (250 mm × 4.6 mm, 3 μm); mobile phase: 60:40 composition of supercritical CO2 and MeOH with 0.1 or 2% formic acid; flow rate: 3 mL/min; detection mode: Agilent DAD UV-detector [Citation126].
![Figure 6. Comparison of chromatograms of FMOC-DL-Alanine-OH in RP-HPLC and SFC. Experimental conditions: column: Lux™ (250 mm × 4.6 mm, 3 μm); mobile phase: 60:40 composition of supercritical CO2 and MeOH with 0.1 or 2% formic acid; flow rate: 3 mL/min; detection mode: Agilent DAD UV-detector [Citation126].](/cms/asset/259f0f39-055f-4b5e-b2e7-aec454c042d0/lspr_a_2038625_f0006_oc.jpg)
Table 2. Achiral separations of amino acids and peptides with SFC using different stationary and mobile phases
Figure 7. Effects of ammonium acetate, trifluoroacetic acid, and water in the mobile phase on the separation of amino acids by SFC: 1. Leucine, 2. Valine, 3. Gamma Aminobutyric Acid, 4. Phenylalanine, 5. Glycine, 6. Theanine, 7. Threonine, 8. Serine, 9. Glutamine, 10. Asparagine, 11. Histidine. Column: Unitary™ XAmide (250 mm × 4.6 mm, 5 μm); mobile phase: supercritical CO2 with indicated additives: 3 mL/min; 25°C; 100 bar; detection mode: selected ion monitoring mode using the precursor ions. Adapted from [Citation135].
![Figure 7. Effects of ammonium acetate, trifluoroacetic acid, and water in the mobile phase on the separation of amino acids by SFC: 1. Leucine, 2. Valine, 3. Gamma Aminobutyric Acid, 4. Phenylalanine, 5. Glycine, 6. Theanine, 7. Threonine, 8. Serine, 9. Glutamine, 10. Asparagine, 11. Histidine. Column: Unitary™ XAmide (250 mm × 4.6 mm, 5 μm); mobile phase: supercritical CO2 with indicated additives: 3 mL/min; 25°C; 100 bar; detection mode: selected ion monitoring mode using the precursor ions. Adapted from [Citation135].](/cms/asset/4b94e51d-6674-41bd-90bf-75baedc22523/lspr_a_2038625_f0007_oc.jpg)
Table 3. Advantages and disadvantages of SFC over other separation techniques in the separation of amino acids and peptides .[Citation80,Citation81,Citation146,Citation147].