Figures & data
Table 1. Summaries of the studies.
Table 2. Natalizumab exposure levels.
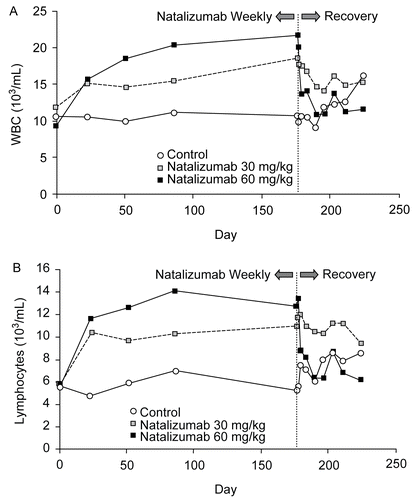
Table 3. Mean change in white blood cell (WBC) and lymphocyte counts in studies of cynomolgus and rhesus monkeys receiving natalizumab.
Figure 2. Phenotype analysis of circulating leukocytes in Study 4 by (A) percentages and (B) absolute counts of cellular subsets.
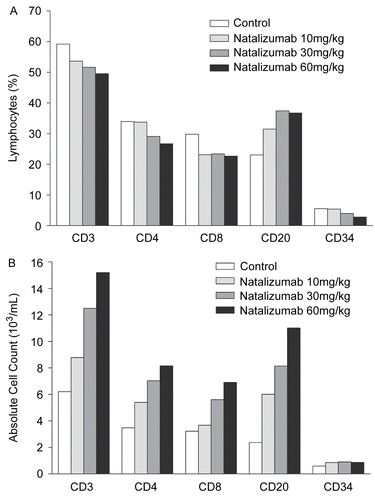
Table 4. Summary of phenotyping of WBC populations in monkeys treated with natalizumab.
Table 5. Results of immune function tests in lymphocytes and spleen cells obtained from cynomolgus monkeys in Study 2 that were treated with natalizumab 30 mg/kg every other day for 4 weeks.
Figure 3. (A) IgM and (B) IgG responses to immunization with keyhole limpet hemocyanin (KLH; Days 7, 35, and 63) and tetanus toxoid (TT; Days 56 and 70) in Study 5. Closed symbols/solid lines, anti-KLH antibodies (center-point titer); open symbols/dotted lines, anti-TT antibodies (cut-point titer); circles, control; triangles, natalizumab 3 mg/kg; squares, natalizumab 30 mg/kg; *p < 0.05 vs. control.
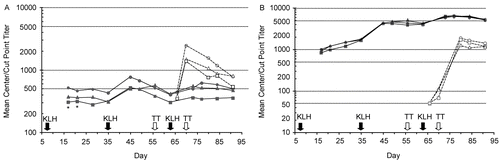
Table 6. Anti-KLH and anti-TT titers in monkeys treated with natalizumab.
Table 7. Results of in vitro immune function tests in PBMC and monocytes from healthy human volunteers (N=5).